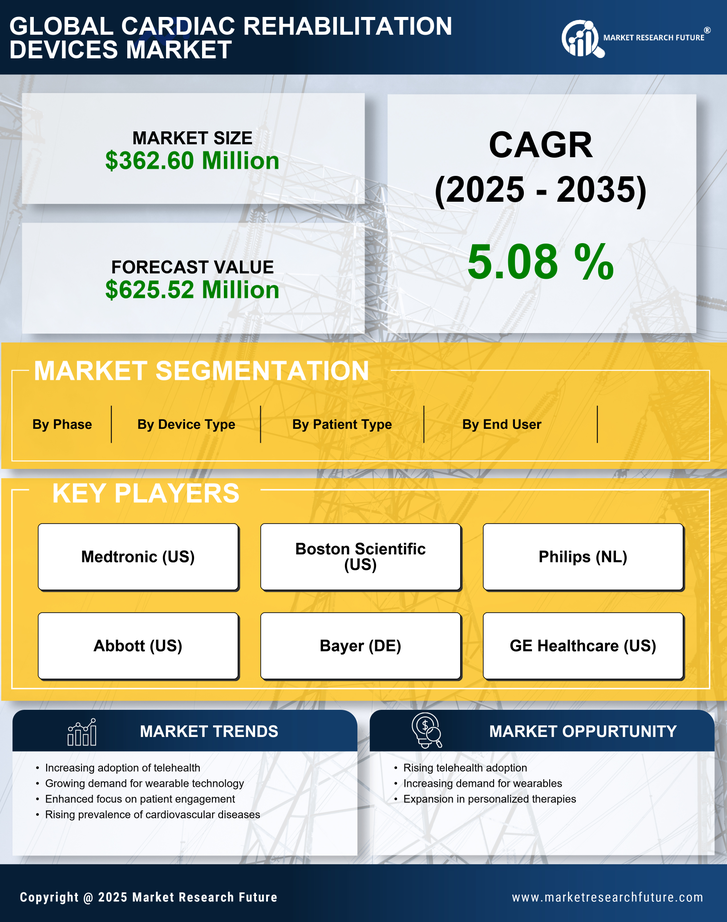
US Medical Device Market Segmentation
Medical Device By Device Type (USD Million, 2025-2035)
- Elliptical Trainer
- Rower
- Blood Pressure Monitor
- Treadmill
- Stabilization Ball
- Heart Rate Monitor
Medical Device By End User (USD Million, 2025-2035)
- Rehab Centers
- Research Centers
- Specialty Centers
- Hospitals
- Clinics
Medical Device By Patient Type (USD Million, 2025-2035)
- Percutaneous Coronary Intervention
- Myocardial Ischemia
- Coronary Artery Bypass Grafting
- Coronary Artery Disease
Medical Device By Phase (USD Million, 2025-2035)
- Phase I
- Phase II
- Phase III