US Alpha Thalassemia Market
ID: MRFR/LS/17741-HCR
100 Pages
Vikita Thakur
Last Updated: April 06, 2026
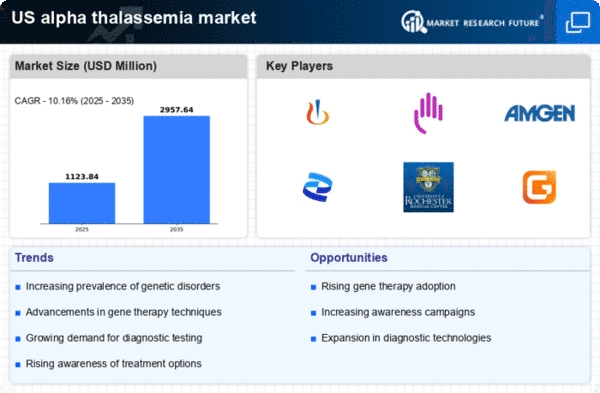
US Alpha Thalassemia Market Research Report By Type (hemoglobin Bart hydrops fetalis syndrome or Hb Bart syndrome, HbH disease, silent carrier state, trait), By Diagnosis (perinatal testing, prenatal testing, pre-implantation, complete blood count (CBC), DNA testing, prenatal genetic testing, chorionic villus sampling), By Treatment (blood transfusions, iron chelation, bone marrow or stem cell transplant, surgery, gene therapy) and By End-User (hospitals, biotechnological laboratories, diagnostic laboratories, educational research institutes, pharmaceutical industries, others) - Growth & Industry Forecast 2025 To 2035
.png)