US Acoustic Neuroma Market
ID: MRFR/MED/17442-HCR
100 Pages
Satyendra Maurya
Last Updated: April 06, 2026
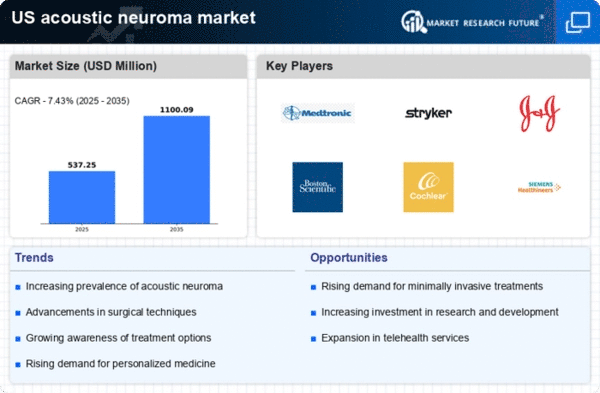
US Acoustic Neuroma Market Research Report By Type (unilateral vestibular schwannomas, bilateral vestibular schwannomas), By Diagnosis (hearing test (audiometry), electronystagmography, magnetic resonance imaging (MRI), others), By Drug Class (anticonvulsants, corticosteroids, osmotic diuretics, pain killers), By Treatment (monitoring, surgery, radiation therapy, supportive therapy, others, stereotactic radiotherapy, proton beam therapy, radiosurgery, craniotomy, microsurgery removal) and By End User (hospitals, diagnostic centers, academic and research institutes, others) - Growth & Industry Forecast 2025 To 2035