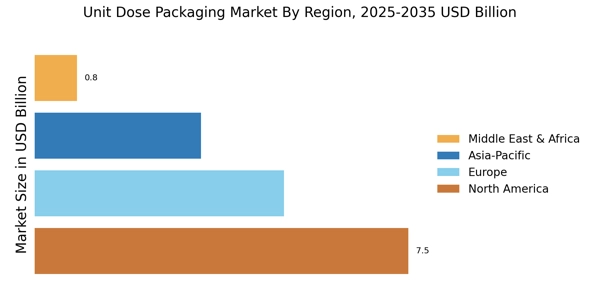
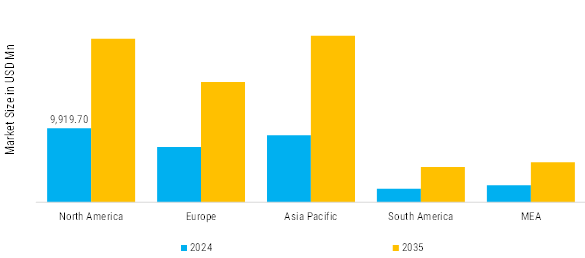
North America: Expanding clinical environment
In North America, expectations surrounding the clinical environment value sterility, traceability, and regulatory compliance highly. Hospital inpatient-feeding and specialty clinic outpatient infusion care patients are generating safe demand for pre-filled syringes, single-use ampoules, and sterile unit vials that serve to reduce medication errors and enhance workflow efficiency in environments dealing with increasingly higher levels of staff shortages. This occurs alongside the growth of vaccination and biologics therapy programs that are reliant on validated cold-chain capabilities and container-closure integrity testing reliability within the organizations' distributed fulfillment systems.
Europe: Strong Production clinical environment
The United Kingdom's unit dose framework is influenced by an array of advanced clinical demand, effective public health systems and increasingly strict environmental regulations which influence the choice of materials. Current Eurostat reports for 2022 indicate that per capita healthcare costs in the EU average approximately €3685, with approximate total current healthcare expenditures of €1648 billion—European figures position the EU as a sophisticated consumer of sterile medical packaging and further consumer-facing unit dose approaches (PCA, single-serves).
Asia Pacific: Fastest Growing clinical environment
Asia-Pacific regions, the workforce capacity, and health system readiness, also require constraints to be considered. For example, even though tertiary centres might be well staffed, and have high high-tech innovation adoption, many lower income areas might be tripled staffed to ensure those identifiable patients are vaccinated or treated. Some locations in Asia-Pacific boast substantial clinical sophistication while enormous and rising mass markets with very low or relatively low or rising income exist in other markets. The OECD data shows there is substantial per capita health expenditure increases; low- and lower-middle income countries have seen the largest inflation adjusted increases in the previous decade. Despite this development, there are substantial differences between them, and so the associated product strategies should be developed and managed by country, and then customers.
South America: Growing clinical environment
The health systems in South America are characterized by strong public programs in a few nations, mixed funding frameworks and recurrent pressure from outbreaks of infectious diseases and climate-related shocks — all of which drive demand for resilient unit dose packaging. Recent reporting in the region and work from the OECD, underscore that Latin America and the Caribbean are under ongoing pressure on health financing and workforce capacity despite improved per capita expenditure across varying national levels; emphasis on public investment targets and improved primary care networks are clearly key priorities.
Middle East & Africa: Emerging clinical environment
The Middle East and Africa have a challenging reality. Some countries are experiencing a rapid rise in healthcare investment while others face an acute fragility of the system. In particular, work by WHO on global health workforce indicates a health worker shortage of approximately 11 million by 2030, the global shortage is concentrated in low- and lower-middle-income countries—a structural constraint that is impacting how unit dose packaging is chosen and utilized through the region.