ID: MRFR/Pharma/51191-HCR
200 Pages
Satyendra Maurya
Last Updated: February 06, 2026
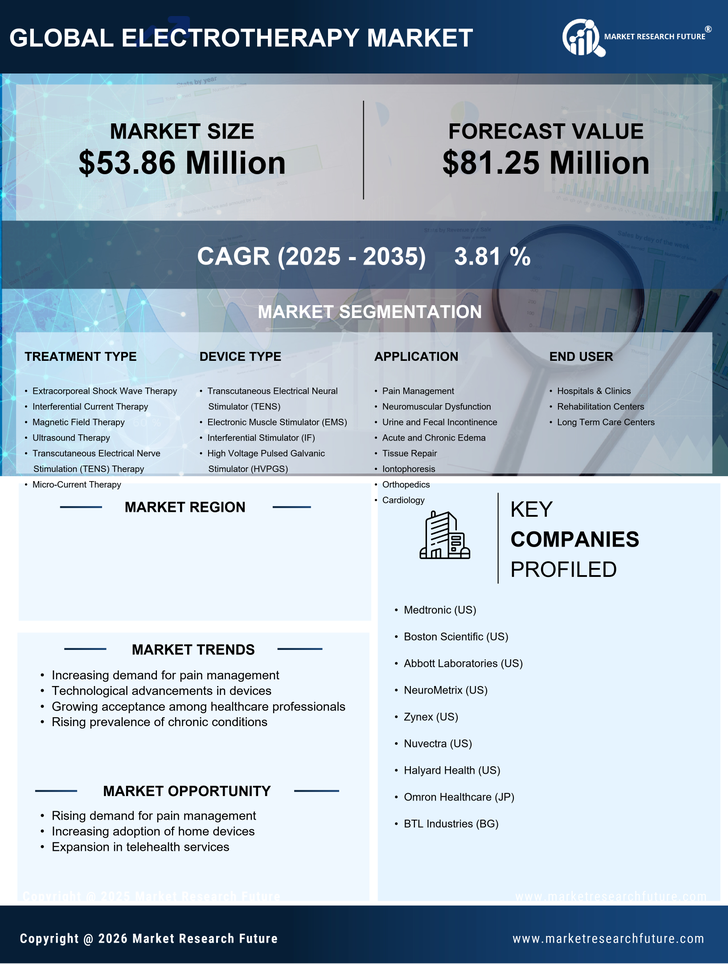
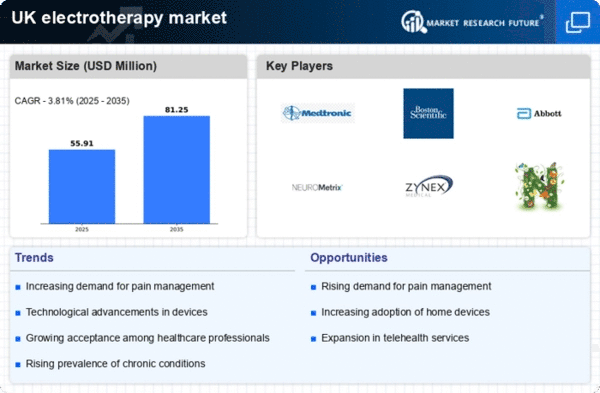
UK Electrotherapy Market Research Report By Treatment Type (Extracorporeal Shock Wave Therapy, Interferential Current Therapy (IC), Magnetic Field Therapy, Ultrasound Therapy, Transcutaneous Electrical Nerve Stimulation (TENS) Therapy, Micro-Current Therapy), By Device Type (Transcutaneous Electrical Neural Stimulation (TENS), Electronic Muscle Stimulator (EMS), Interferential Stimulator (IF), High Voltage Pulsed Galvanic Stimulator (HVPGS)), By Application (Pain Management, Neuromuscular Dysfunction, Urine and Fecal Incontinence, Acute and Chronic Edema, Tissue Repair, Iontophoresis, Orthopedics, Cardiology) and By End User (Hospitals and amp; Clinics, Rehabilitation Centers, Long Term Centers) -Forecast to 2035