Market Trends
Key Emerging Trends in the Tyrosine Hydroxylase Deficiency Market
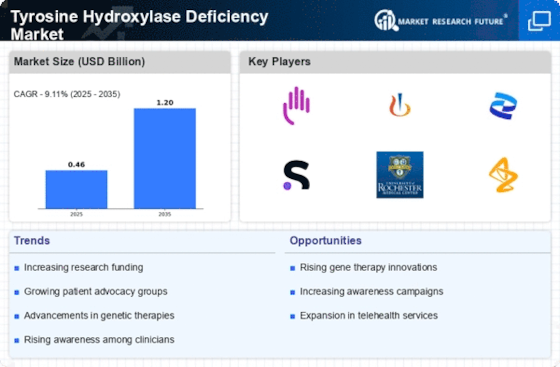
A complex landscape exists in the market trends of Tyrosine Hydroxylase Deficiency (THD) driven by advancements in medical research, better diagnostic capability and an increasing understanding of rare genetic disorders. THD is a rare inherited disorder that affects neurotransmitter synthesis causing difficulties in diagnosis and management. One notable trend is the increased global recognition of THD due to better awareness amongst healthcare providers and improved genetics testing. The increasing awareness plays a part in early detection and intervention which is crucial in what could be considerable neurological consequences of THD. The move towards earlier identification also reflects a broader shift within the field of rare disease research toward addressing unmet needs with an aim to improve patient outcomes.
Among all these, there are developments made in diagnostic tools such as genetic tests and molecular analysis for early diagnostics for THD. Genetic mutations specific for THD make it possible for the doctors to confirm the diagnosis as well as comprehend its underlying genetic basis. This trend towards more accurate diagnostic capabilities ensures prompt intervention and enables targeted treatment strategies for people affected by Tyrosine Hydroxylase Deficiency.
The pharmaceutical industry has shown increased interest on rare diseases leading to the initiation of research activities aimed at addressing THD. The neurochemical imbalances associated with this condition are redressed through therapeutic approaches because THD is a disorder that affects production of neurotransmitters like dopamine. Although treatments specifically for THD may still be at their infancy stages, ongoing studies show potential breakthroughs targeting molecular mechanisms that underlie this condition. This surge highlights need for advancement in treatment options available for patients having rarer genetic ailments like THD.
With reference to Tyrosine Hydroxylase Deficiency, personalized medicine has increasingly influenced market trends by health organizations. Treatment plans are now customized based on individual patient characteristics, including specific gene mutations and symptom severity. This precision medicine aims at increasing its therapeutic efficiency while reducing possible side effects thus making it more patient-centered as well as enhancing management of THD.
Collaborations among healthcare providers, researchers, pharmaceutical companies and patient advocacy groups are influencing the landscape of THD care. The establishment of specialized centers of excellence and research networks ensures a comprehensive and coordinated approach to patient care. These collaborations support the conduct of clinical trials, research initiatives, and sharing knowledge about Tyrosine Hydroxylase Deficiency.
Telemedicine and digital health technologies have become essential aspects of THD care by facilitating remote consultations, genetic counseling as well as providing ongoing support for affected persons including their families. Such implementations enhance accessibility especially for patients residing in geographically distant areas while creating a platform for knowledge exchange among health practitioners involved in rare genetic disorders. Telemedical integration supports wider adoption of digital health applications that provide easy management solutions like in case with rare diseases such as THD.
The potential for gene therapy and emerging treatment modalities is an exciting trend in the THD market. Although gene therapy approaches towards treating THD may still be experimental, there is hope that underlying genetic mutations could be addressed through this mechanism. Continuing studies on gene editing technologies coupled with advances made within the genetic medicine field might lead to radical treatments for Tyrosine Hydroxylase Deficiency at some point in time.


















Leave a Comment