Market Trends
Key Emerging Trends in the Thrombus Treatment Market
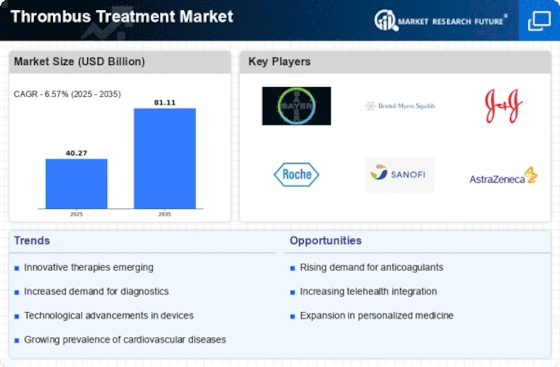
The thrombus treatment market is witnessing dynamic trends that reflect the evolving landscape of healthcare, advancements in medical research, and a growing emphasis on patient-centric care. These trends are shaping the industry, influencing treatment approaches, and contributing to the development of innovative solutions. Let's explore some key trends in the thrombus treatment market.
Advancements in Anticoagulant Therapies: A notable trend in the thrombus treatment market is the continuous advancement of anticoagulant therapies. Research and development efforts are focused on refining existing anticoagulant drugs and developing novel options with improved efficacy and safety profiles. The goal is to offer more targeted and personalized treatment options for patients with thrombus-related conditions.
Rise of Oral Anticoagulants: Oral anticoagulants have gained prominence as a preferred mode of treatment for thrombus-related disorders. The convenience of oral administration, coupled with comparable effectiveness to traditional injectable anticoagulants, has contributed to the increasing adoption of oral anticoagulant therapies. This trend aligns with the broader shift towards patient-friendly treatment modalities.
Personalized Medicine and Biomarkers: The era of personalized medicine is making inroads into thrombus treatment. Researchers are exploring biomarkers and genetic factors to tailor treatment plans based on individual patient characteristics. This personalized approach aims to optimize therapeutic outcomes while minimizing side effects, marking a significant shift towards more precise and patient-specific interventions.
Technological Integration in Treatment Devices: Advancements in technology are influencing treatment devices used in thrombus management. Innovations such as smart catheters and improved thrombectomy systems are enhancing the precision and effectiveness of interventional procedures. This trend reflects a commitment to leveraging technology for better patient outcomes and procedural success.
Telemedicine and Remote Monitoring: The adoption of telemedicine and remote monitoring is transforming the follow-up and management of thrombus patients. Remote monitoring solutions enable healthcare providers to track patients' progress, adjust treatment plans, and provide timely interventions, enhancing the continuity of care and improving patient outcomes.
Focus on Preventive Strategies: There is a growing emphasis on preventive strategies to mitigate the risk of thrombus formation. Lifestyle interventions, education on early warning signs, and the development of prophylactic medications contribute to a holistic approach in preventing thrombus-related complications. This trend aligns with the broader shift towards preventive healthcare.
Patient Education and Awareness Programs: Recognizing the importance of patient awareness, there is an increasing focus on education programs aimed at informing individuals about thrombus risk factors, symptoms, and preventive measures. Empowering patients with knowledge enhances early detection and encourages proactive management, fostering better overall health outcomes.
Global Collaborations in Research: Collaborations among research institutions, healthcare organizations, and pharmaceutical companies on a global scale are accelerating advancements in thrombus treatment. The pooling of resources, expertise, and data facilitates more robust research outcomes and the development of innovative therapies. This collaborative trend reflects a commitment to addressing thrombus-related challenges collectively.
Integration of Artificial Intelligence (AI) in Diagnosis: The integration of artificial intelligence in diagnostic processes is gaining traction in thrombus treatment. AI-powered algorithms analyze imaging data with high precision, aiding in early and accurate thrombus detection. This technological trend enhances diagnostic efficiency and contributes to timely interventions.
Environmental Sustainability in Treatment Options: There is an emerging trend towards environmental sustainability in the development and production of thrombus treatment options. Companies are exploring eco-friendly practices, from sourcing materials to manufacturing, aligning with broader initiatives for sustainable and responsible healthcare practices.
The thrombus treatment market is undergoing transformative changes driven by advancements in treatment modalities, technology, and a heightened focus on patient-centric care. These trends collectively contribute to a more nuanced and effective approach to managing thrombus-related conditions, reflecting the commitment of the healthcare industry to continuous innovation and improvement in patient outcomes. As research and technological advancements continue, the thrombus treatment landscape is poised for further evolution, offering new possibilities for enhanced care and better quality of life for patients.



















Leave a Comment