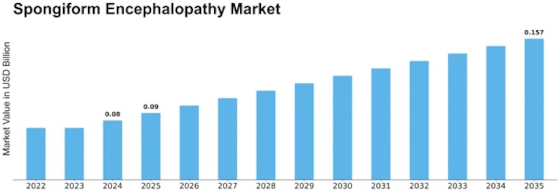
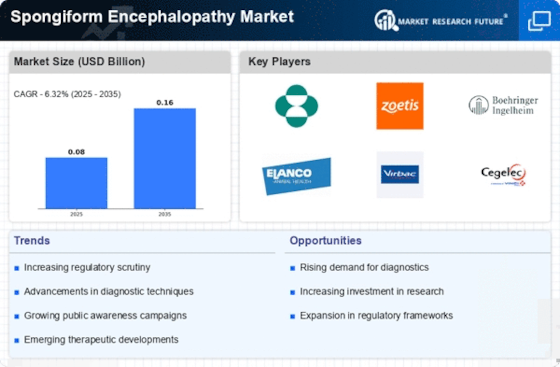
Spongiform Encephalopathy Size
Spongiform Encephalopathy Market Growth Projections and Opportunities
The market is essentially affected by the predominance and frequency paces of Spongiform Encephalopathy. The quantity of detailed cases, both universally and locally, directly influences the interest for analytic instruments, treatment choices, and steady consideration, driving market growth. Progressions in demonstrative advancements assume a critical part in the Spongiform Encephalopathy market. Improved and more exact analytic instruments upgrade early identification, empowering convenient intercession and management. The market answers decidedly to developments in biomarker identification proof, imaging procedures, and genetic testing. Progressing innovative work drives add to market extension. Interests in figuring out the hidden causes, creating novel helpful methodologies, and improving patient consideration lift the market by offering new arrangements and treatment modalities. Financial variables, both worldwide and territorial, influence the Spongiform Encephalopathy market. Financial steadiness, medical care framework advancement, and government medical services spending influence openness to demonstrative instruments and therapy choices, impacting market elements. The presentation of imaginative treatment modalities essentially shapes the market. Forward leaps in drug improvement, quality treatments, and other treatment approaches add to the extension of accessible choices for patients, cultivating rivalry among drug organizations. Protection inclusion and repayment contracts influence patient admittance to demonstrative tests and medicines. Good repayment strategies can animate market growth by guaranteeing that medical care suppliers are satisfactorily made up for their administrations, empowering interest in Spongiform Encephalopathy management. Segment patterns, like a maturing population, can impact the market. With Spongiform Encephalopathy being more predominant in more seasoned people, segment shifts add to an expanded patient pool, influencing the interest for related medical care administrations and items.



















Leave a Comment