North America : Market Leader in Innovation
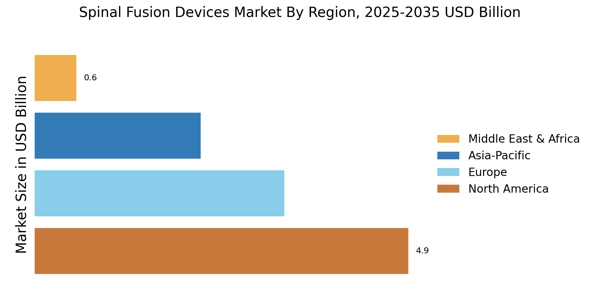
North America leads the Spinal Fusion Devices Market, accounting for 45% of the global revenue in 2024. The region's growth is driven by an aging population, increasing prevalence of spinal disorders, and advancements in surgical techniques. Regulatory support from agencies like the FDA has also catalyzed innovation and spine product approvals, enhancing market dynamics. The demand for minimally invasive procedures is further propelling market growth, as patients seek quicker recovery times and reduced hospital stays.
The United States is the primary contributor to this market, with key players such as Medtronic, DePuy Synthes, and Stryker leading the competitive landscape. The presence of advanced healthcare infrastructure and significant investment in research and development fosters a robust environment for innovation. Additionally, the growing trend of outpatient surgeries is reshaping the market, as hospitals and clinics adopt new technologies to improve patient outcomes.
Europe : Emerging Market with Growth Potential
Europe Spinal Fusion Devices Market accounted for 30% share in 2024, generating around USD 3.276 billion, making it the second-largest regional market. The region's growth is fueled by increasing healthcare expenditure, a rise in spinal surgeries, and a growing elderly population. Regulatory frameworks, such as the Medical Device Regulation (MDR), are enhancing spine product safety and efficacy, which in turn boosts consumer confidence and market demand.
Countries like Germany and France are leading the charge, with significant investments in healthcare infrastructure. Germany stands out as a key player in the European market, with a strong presence of companies like B. Braun and Zimmer Biomet. The competitive landscape is characterized by a mix of established firms and emerging startups, fostering innovation and diverse spine product offerings. Collaborative efforts between healthcare providers and manufacturers are also on the rise, aimed at improving surgical outcomes and patient care. This dynamic environment positions Europe as a promising market for future growth.
Asia-Pacific : Rapidly Growing Market Segment
Asia-Pacific is witnessing rapid growth in the spinal fusion devices market, accounting for approximately 20% of the global share. The region's expansion is driven by rising healthcare awareness, increasing incidences of spinal disorders, and improvements in healthcare infrastructure. Countries like China and India are experiencing a surge in demand for advanced medical technologies, supported by government initiatives aimed at enhancing healthcare access.
The growing trend of medical tourism in these countries is also contributing to market growth. China is emerging as a significant player in this market, with local manufacturers and international companies like Globus Medical and Orthofix expanding their presence. The competitive landscape is evolving, with a focus on affordability and accessibility of spinal fusion devices. Additionally, partnerships between healthcare providers and device manufacturers are fostering innovation, ensuring that the region keeps pace with global advancements in spinal surgery technologies.
Middle East and Africa : Untapped Market Potential
The Middle East and Africa (MEA) region is gradually emerging in the spinal fusion devices market, holding about 5% of the global share. The growth is primarily driven by increasing healthcare investments, a rising prevalence of spinal disorders, and a growing awareness of advanced surgical options. Governments in the region are focusing on improving healthcare infrastructure and access, which is expected to catalyze market growth.
The demand for spinal fusion procedures is on the rise, particularly in countries like South Africa and the UAE, where healthcare systems are evolving. South Africa is leading the market in the MEA region, with a growing number of hospitals adopting advanced spinal technologies. The competitive landscape is characterized by a mix of local and international players, with companies like K2M and Stryker establishing a foothold. The region's potential for growth is significant, as healthcare providers increasingly seek innovative solutions to meet the rising demand for spinal surgeries and improve patient outcomes.