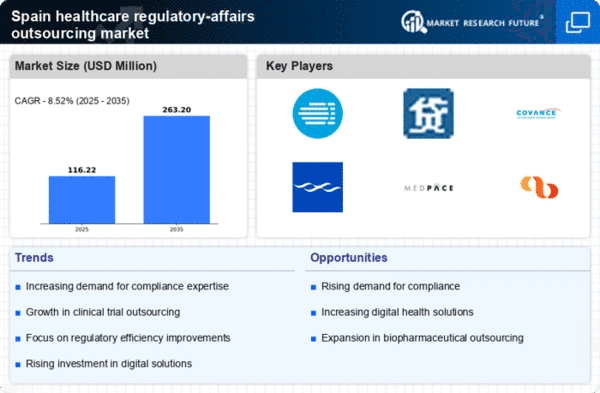
Spain Life Sciences Market Segmentation
Life Sciences By Service (USD Million, 2025-2035)
- Regulatory Writing and Publishing
- Regulatory Submissions
- Clinical Trial Applications and Services Registrations
- Regulatory Consulting and Legal Representation
- Other Regulatory Affairs
Life Sciences By End User (USD Million, 2025-2035)
- Mid-Size Pharmaceutical Companies
- Large Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Food and Beverage Companies