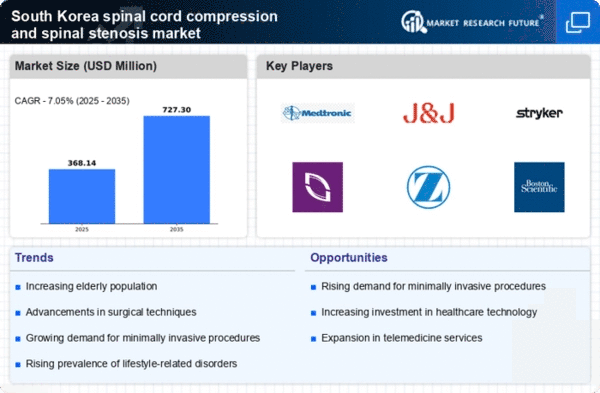
South Korea Medical Device Market Segmentation
Medical Device By Type (USD Million, 2025-2035)
- Cervical Stenosis
- Central Stenosis
- Lumbar Spinal Stenosis
Medical Device By Diagnosis (USD Million, 2025-2035)
- Physical Examination
- Imaging
Medical Device By Treatment Type (USD Million, 2025-2035)
- Surgical Treatment
- Non-Surgical Treatment
Medical Device By End-Users (USD Million, 2025-2035)
- Ambulatory Surgical Centers
- Rehabilitation Centers
- Hospitals