South Korea Control Valve Market
ID: MRFR/EnP/46398-HCR
128 Pages
Chitranshi Jaiswal
Last Updated: April 21, 2026
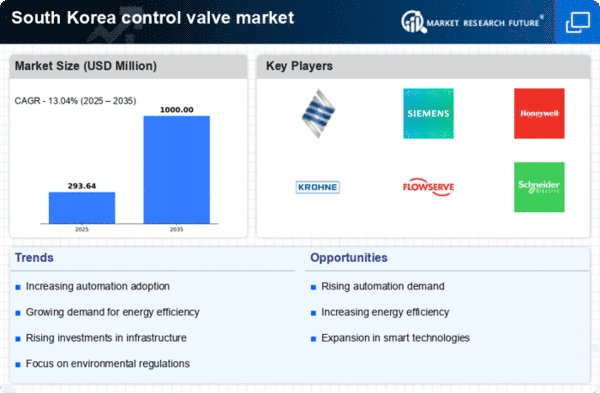
South Korea Control Valve Market Research Report By Valve Type (Rotary valves, Linear valves), By Valve Size (Below 1”, 1” To 6”, 25” To 50”, Above 50”), By Material (Stainless Steel, Cast Iron, Alloy-based, Cryogenic, Others), By Component (Valve Body, Actuator, Others) andBy End-Use (Oil & Gas, Water & Wastewater Treatment, Energy & Power, Chemicals, Food & Beverage, Pharmaceuticals, Others) - Growth & Industry Forecast to 2035