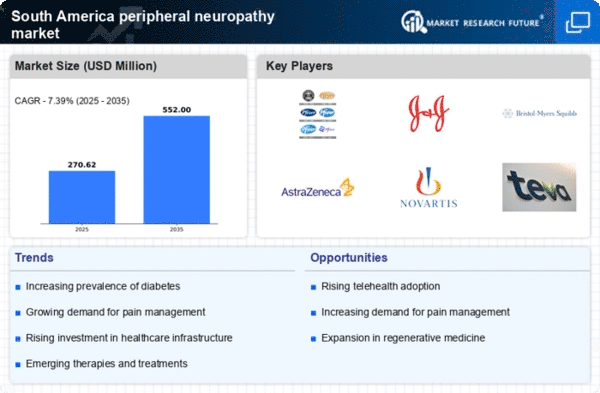
South America Life Sciences Market Segmentation
Life Sciences By Type (USD Million, 2025-2035)
- Diabetic Peripheral Neuropathy
- Chemotherapy-induced Peripheral Neuropathy
- Idiopathic Peripheral Neuropathy
- HIV/AIDS Associated Peripheral Neuropathy
Life Sciences By Treatment (USD Million, 2025-2035)
- Pharmacological Therapies
- Non-Pharmacological Therapies
- Others
Life Sciences By End User (USD Million, 2025-2035)
- Hospitals and Clinics
- Ambulatory Centers
- Others