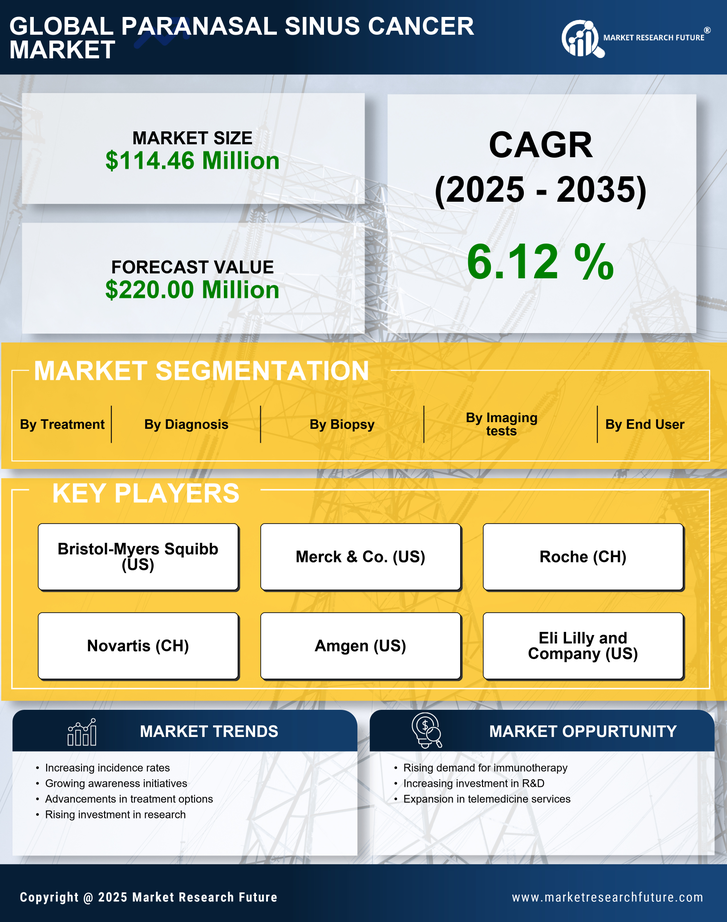
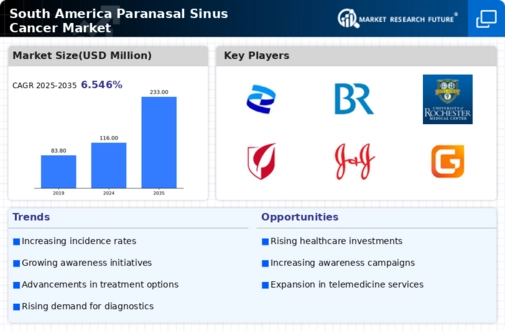
South America Pharmaceutical Market Segmentation
Pharmaceutical By Diagnosis (USD Million, 2025-2035)
- Medical history and physical examination
- Imaging tests
- Biopsy
- Others
Pharmaceutical By Imaging tests (USD Million, 2025-2035)
- X-rays
- CT (computed tomography) scan
- MRI (magnetic resonance imaging) scan
- PET (positron emission tomography) scan
Pharmaceutical By Biopsy (USD Million, 2025-2035)
- Fine needle aspiration (FNA) biopsy
- Incisional and excisional biopsies
Pharmaceutical By Treatment (USD Million, 2025-2035)
- Surgery
- Radiotherapy
- Chemotherapy
Pharmaceutical By End User (USD Million, 2025-2035)
- Hospital & Clinics
- Cancer Treatment & Research Center
- Research Laboratories
- Others