South America Interventional Radiology Products Market
ID: MRFR/HC/49721-HCR
200 Pages
Rahul Gotadki
Last Updated: February 06, 2026
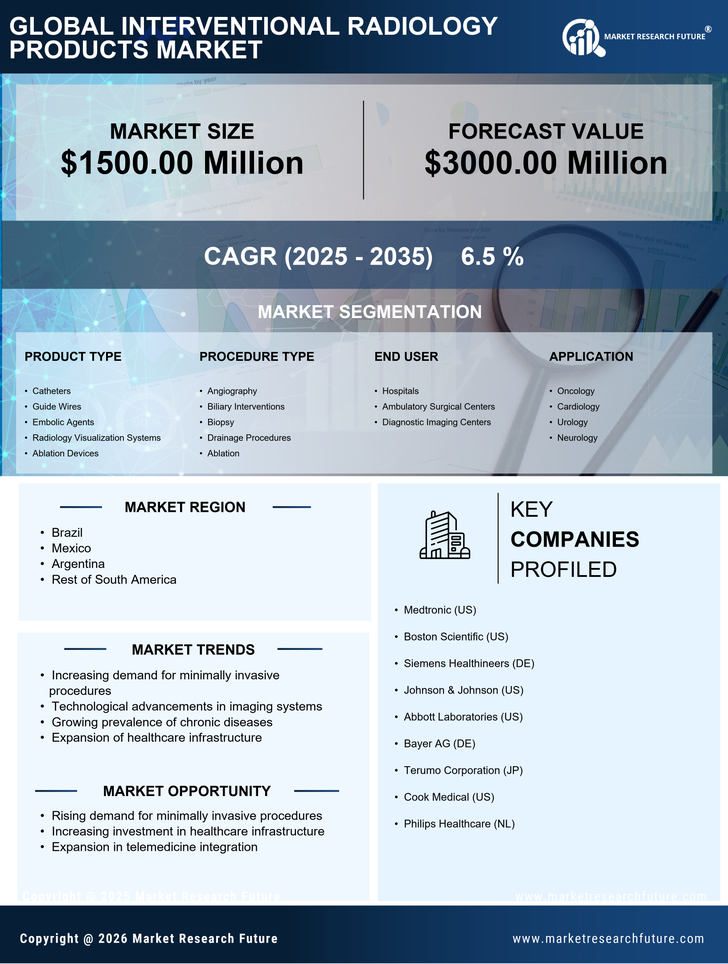
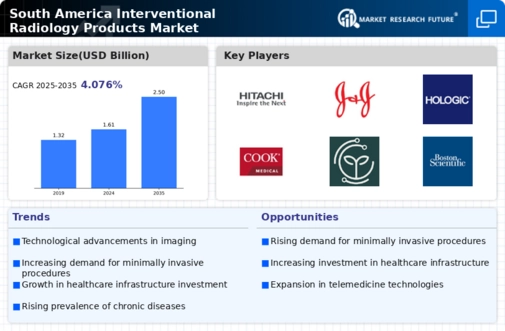
South America Interventional Radiology Products Market Research Report By Product Type (Catheters, Guide Wires, Embolic Agents, Radiology Visualization Systems, Ablation Devices), By Procedure Type (Angiography, Biliary Interventions, Biopsy, Drainage Procedures, Ablation), By End User (Hospitals, Ambulatory Surgical Centers, Diagnostic Imaging Centers), By Application (Oncology, Cardiology, Urology, Neurology) and By Regional (Brazil, Mexico, Argentina, Rest of South America) - Growth & Industry Forecast 2025 To 2035