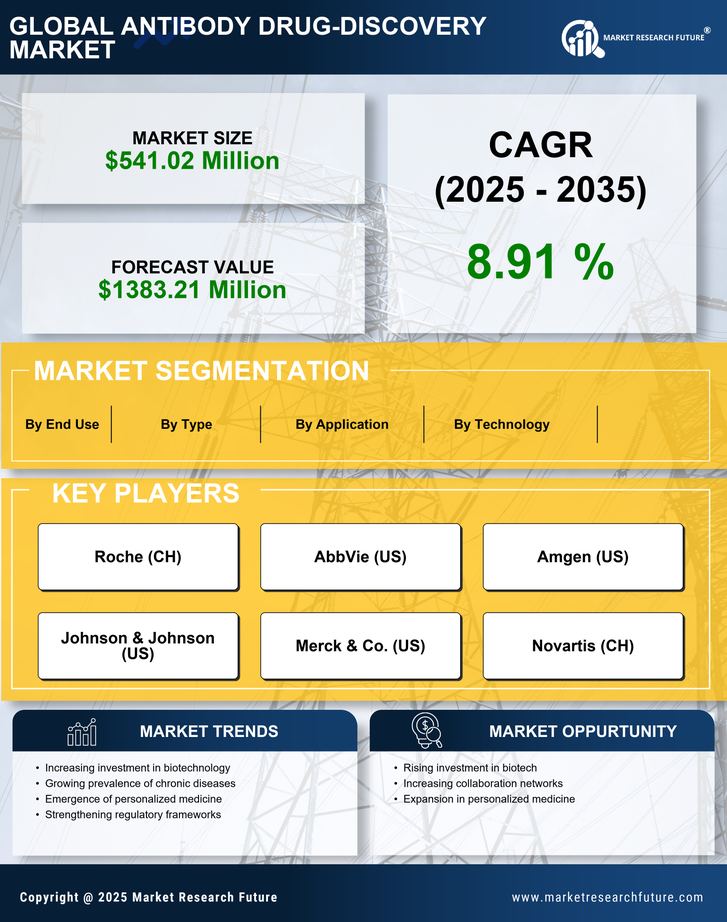
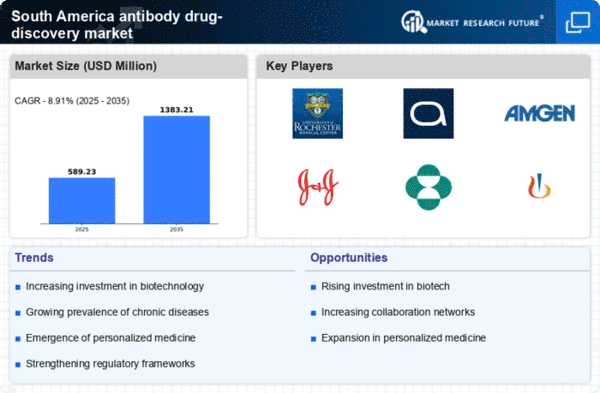
South America Security, Access Control and Robotics Market Segmentation
Security, Access Control and Robotics By Type (USD Million, 2025-2035)
- Monoclonal Antibodies
- Polyclonal Antibodies
- Antibody-Drug Conjugates
- Bispecific Antibodies
Security, Access Control and Robotics By Technology (USD Million, 2025-2035)
- Recombinant DNA Technology
- Hybridoma Technology
- Phage Display Technology
- Transgenic Technology
Security, Access Control and Robotics By Application (USD Million, 2025-2035)
- Therapeutic Applications
- Diagnostic Applications
- Research Applications
Security, Access Control and Robotics By End Use (USD Million, 2025-2035)
- Pharmaceutical Companies
- Biotechnology Companies
- Research Institutions