Refurbished Medical Devices Market Summary
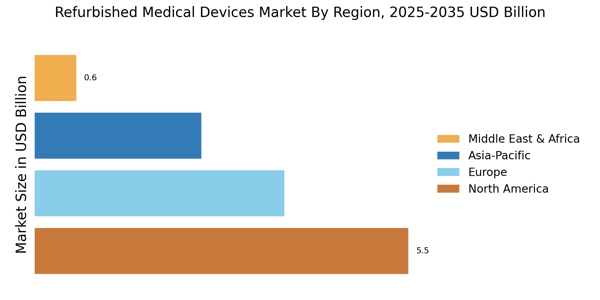
According to Market Research Future analysis, the Refurbished Medical Devices Market Size was valued at USD 12.33 Billion in 2024 & the market is projected to grow from USD 13.27 Billion in 2025 to USD 27.69 Billion by 2035, registering a CAGR of 7.63% during the forecast period 2025–2035. North America led the market with over 45% share, generating around USD 5.55 billion in revenue. The market is primarily driven by rising healthcare cost pressures and the need for affordable diagnostic and treatment solutions. Healthcare providers increasingly adopt refurbished equipment to expand access while maintaining quality care standards and operational efficiency globally.
According to World Health Organization, global healthcare spending reached over USD 9.8 trillion, accounting for nearly 10% of GDP, encouraging cost-efficient solutions like refurbished devices. Additionally, Institute for Health Metrics and Evaluation highlights increasing healthcare demand, with global disease burden rising significantly, reinforcing adoption of affordable medical technologies across emerging and developed healthcare systems.
Key Market Trends & Highlights
The Refurbished Medical Devices Market is experiencing robust growth driven by sustainability and cost efficiency.
- North America holds over 45% market share in 2024, driven by high healthcare spending and strong regulatory support.
- Europe market reached USD 3.70 billion in 2024, accounting for 30% share with sustainability-driven refurbishment adoption.
- Imaging equipment dominates with 42% share, driven by increasing global diagnostic demand and rising chronic disease prevalence worldwide.
- Hospitals account for 48% share, supported by growing patient volume and need for cost-efficient, high-performance medical equipment solutions.
- Cardiology leads applications with 38% share, fueled by increasing cardiovascular disease burden and demand for affordable diagnostic technologies.
Market Size & Forecast
| 2024 Market Size | 12.33 (Billion USD) |
| 2035 Market Size | 27.69 (Billion USD) |
| CAGR (2025 - 2035) | 7.63% |
Major Players
Companies such as GE Healthcare (US), Philips Healthcare (NL), Siemens Healthineers (DE), Fujifilm Medical Systems (JP), Mindray (CN), Stryker (US), Olympus Corporation (JP), Nihon Kohden Corporation (JP), Interscope Medical (US) are some of the major participants in the global market.