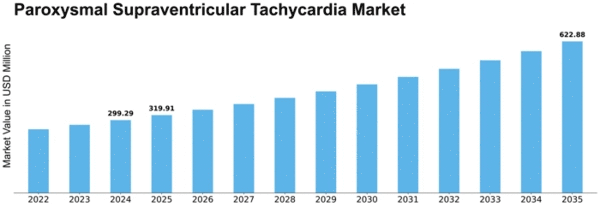
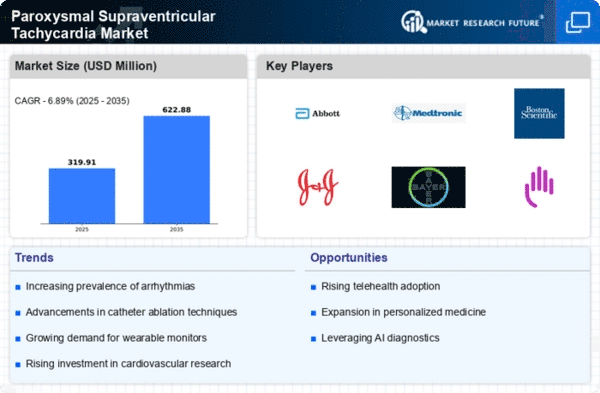
Paroxysmal Supraventricular Tachycardia Size
Paroxysmal Supraventricular Tachycardia Market Growth Projections and Opportunities
The market for paroxysmal supraventricular tachycardia (PSVT) is molded by the meticulousness of this arrhythmia. PSVT includes quick pulses beginning over the heart's ventricles, and its attributes impact the advancement of designated symptomatic and treatment arrangements inside the more extensive arrhythmia market. The market is impacted by progressions in electrophysiological planning and removal procedures well defined for PSVT. Catheter removal techniques, which include designated annihilation of strange tissue causing PSVT, add to the expanding exhibit of treatment choices. Organizations at the cutting edge of refining these methods add to the developing landscape of PSVT management. Drug organizations assume a critical part in the PSVT market by creating hostile to arrhythmic medications to oversee and control episodes. Medications, for example, beta-blockers and calcium channel blockers are commonly recommended. Innovative work endeavors in drug advancement influence market patterns, expecting to upgrade drug adequacy and limit consequences. The connection of telehealth and remote checking arrangements is progressively relevant in the management of PSVT. Remote checking takes into consideration constant following of heart rhythms, empowering healthcare suppliers to quickly intervene. Organizations embracing telehealth add to more accessible and effective healthcare administrations for people with PSVT. Lifestyle factors, like stress, caffeine intake, and absence of physical work, can set off PSVT episodes. The market answers the impact of these variables by stressing way of life alterations as a component of the exhaustive management of PSVT. Organizations elevating all-encompassing methodologies add to a patient-focused market that tends to both intense episodes and long haul prevention. The psychosocial effect of living with PSVT is a huge thought on the lookout. Organizations tending to the close to home and mental parts of the condition, like tension and vulnerability during episodes, add to a more persistent driven approach. Patient help drives upgrade by and large consideration and work on the personal satisfaction for people with PSVT.


















Leave a Comment