Market Trends
Key Emerging Trends in the Mendelian Disorders Testing Market
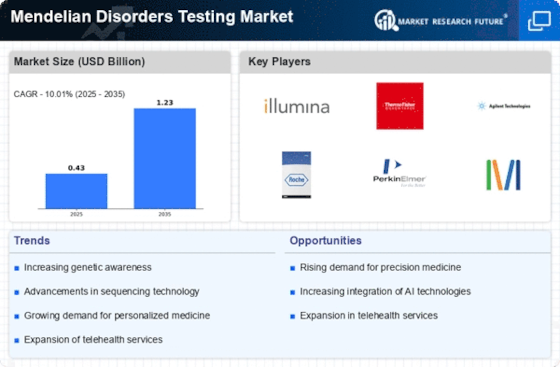
The demand for Mendelian Disorders testing in the United States has been steadily increasing, reflecting a growing recognition of the significance of genetic testing in healthcare. Mendelian Disorders, also known as monogenic disorders, are caused by mutations in a single gene and often have a clear inheritance pattern. This market demand surge can be attributed to various factors, including advancements in genetic testing technologies, heightened awareness of the role of genetics in health, and an increasing prevalence of Mendelian Disorders within the population.
Advancements in genetic testing technologies have played a pivotal role in driving the demand for Mendelian Disorders testing in the US. The field of genomics has witnessed remarkable progress, with the development of high-throughput sequencing technologies, such as next-generation sequencing (NGS). These technologies enable comprehensive analysis of an individual's genetic makeup, allowing healthcare professionals to identify specific gene mutations associated with Mendelian Disorders more efficiently and accurately. As the cost of genetic testing continues to decrease, it has become more accessible to a broader range of individuals, contributing to the growing demand for Mendelian Disorders testing.
There has been a notable increase in public awareness regarding the role of genetics in determining health outcomes. This heightened awareness is driven by factors such as increased media coverage of genetic discoveries, educational initiatives, and the growing popularity of direct-to-consumer genetic testing services. As individuals become more informed about the potential genetic underpinnings of certain health conditions, there is a corresponding increase in demand for Mendelian Disorders testing to assess and understand their own genetic risk factors.
The prevalence of Mendelian Disorders within the population has also contributed significantly to the rising demand for testing services. With an increasing understanding of the genetic basis of various diseases, healthcare professionals are better equipped to identify individuals at risk of Mendelian Disorders and provide targeted genetic testing. This proactive approach to healthcare, focusing on early identification and intervention, has driven the demand for testing services to prevent or manage these disorders effectively.
The US Mendelian Disorders testing market is witnessing a surge in the development and availability of genetic testing panels tailored to specific disorders. These panels allow for the simultaneous analysis of multiple genes associated with a particular Mendelian Disorder, streamlining the testing process and providing comprehensive insights. This trend is especially crucial for conditions with overlapping symptoms, enabling more accurate and timely diagnoses.
In addition to traditional healthcare settings, the demand for Mendelian Disorders testing has extended to research and pharmaceutical sectors. Genetic testing plays a vital role in the identification of potential therapeutic targets and the development of personalized medicine. The collaboration between research institutions, pharmaceutical companies, and genetic testing laboratories has fueled innovation and expanded the scope of Mendelian Disorders testing applications.
As the demand for Mendelian Disorders testing continues to rise, there is a growing emphasis on genetic counseling services to support individuals and families in understanding and interpreting their test results. Genetic counselors play a crucial role in helping individuals make informed decisions about their healthcare based on their genetic information, contributing to a more comprehensive and patient-centered approach to genetic testing.



















Leave a Comment