Medical Device Testing Certification Market
ID: MRFR/MED/41998-HCR
128 Pages
Rahul Gotadki
Last Updated: April 06, 2026
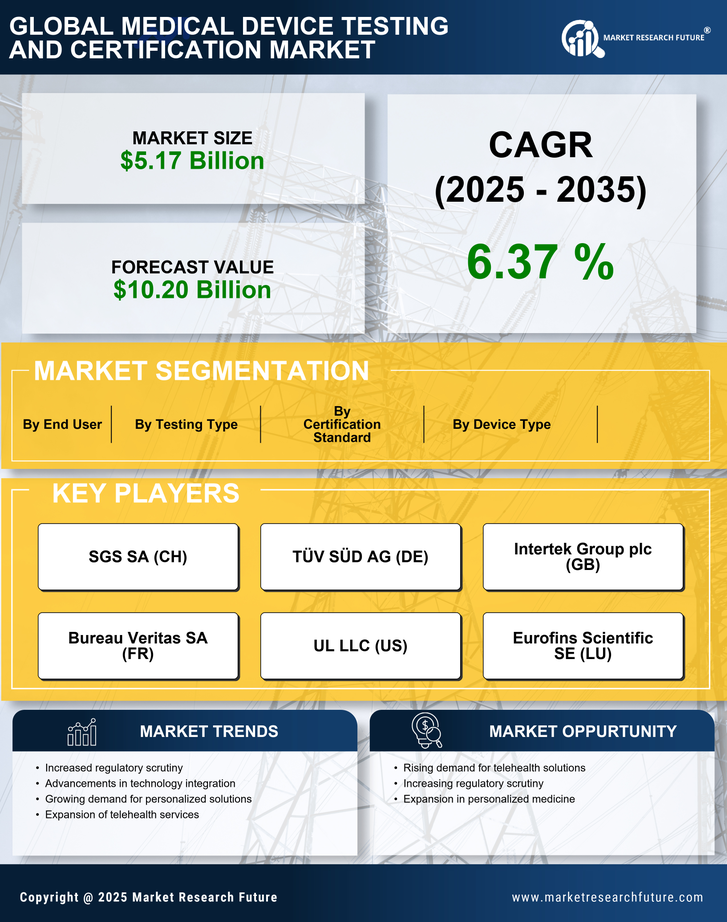
Medical Device Testing and Certification Market Research Report: Size, Share, Trend Analysis By Testing Type (Biocompatibility Testing, Electrical Safety Testing, Performance Testing, Sterilization Validation, Software Testing), By Device Type (In-Vitro Diagnostic Devices, Cardiovascular Devices, Orthopedic Devices, Neurological Devices, Electromedical Devices), By Certification Standard (ISO 13485, IEC 60601, FDA Standards, CE Marking, ISO 14971), By End Users (Medical Device Manufacturers, Healthcare Institutions, Research Organizations, Regulatory Bodies) and By Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth Outlook & Industry Forecast 2025 To 2035