Medical Device Compliance Consulting Market
ID: MRFR/PS/65503-HCR
200 Pages
Rahul Gotadki
Last Updated: March 29, 2026
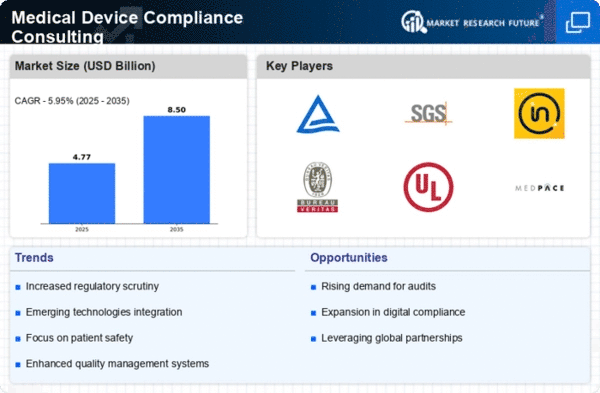
Medical Device Compliance Consulting Research Report Information By End Use (Medical Device Manufacturers, Consulting Firms, Regulatory Authorities, Healthcare Providers, Research Institutions) By Application (Regulatory Compliance, Quality Management Systems, Risk Management, Clinical Evaluation, Post-Market Surveillance) By Device Type (In Vitro Diagnostic Devices, Surgical Instruments, Implantable Devices, Monitoring Devices, Therapeutic Devices) By Type (Pre-Market Consulting, Post-Market Consulting, Compliance Audits, Quality Assurance, Regulatory Strategy Development) By Service Type (Consultation Services, Training And Education, Auditing Services, Documentation Support, Regulatory Submission Support) And By Region (North America, Europe, Asia-Pacific, And Rest Of The World) – Market Forecast Till 2035.