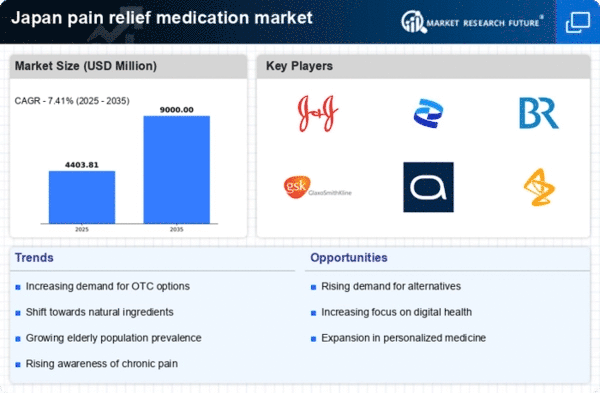
Japan Construction Market Segmentation
Construction By Type (USD Million, 2025-2035)
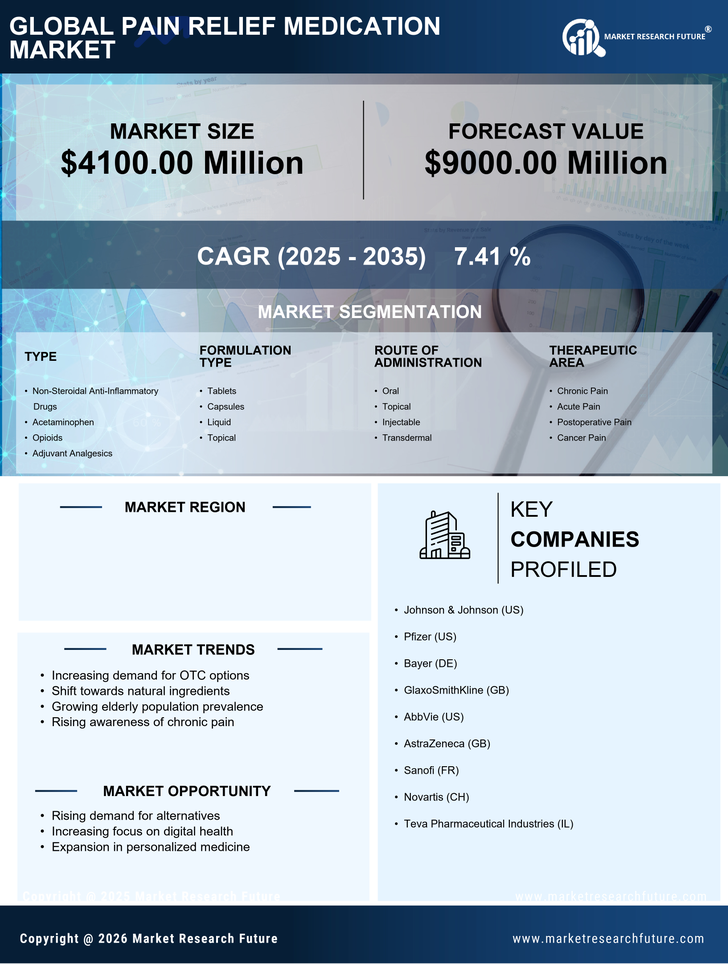
- Non-Steroidal Anti-Inflammatory Drugs
- Acetaminophen
- Opioids
- Adjuvant Analgesics
Construction By Formulation Type (USD Million, 2025-2035)
- Tablets
- Capsules
- Liquid
- Topical
Construction By Route of Administration (USD Million, 2025-2035)
- Oral
- Topical
- Injectable
- Transdermal
Construction By Therapeutic Area (USD Million, 2025-2035)
- Chronic Pain
- Acute Pain
- Postoperative Pain
- Cancer Pain