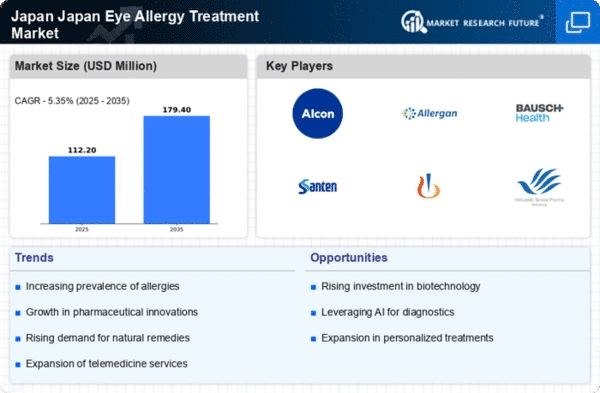
Japan Healthcare Market Segmentation
Healthcare By Treatment Type (USD Million, 2026-2035)
- Antihistamines
- Decongestants
- Mast Cell Stabilizers
- Corticosteroids
Healthcare By Route of Administration (USD Million, 2026-2035)
- Topical
- Oral
- Injectable
Healthcare By Patient Demographics (USD Million, 2026-2035)
- Children
- Adults
- Elderly
Healthcare By Severity of Condition (USD Million, 2026-2035)
- Mild
- Moderate
- Severe