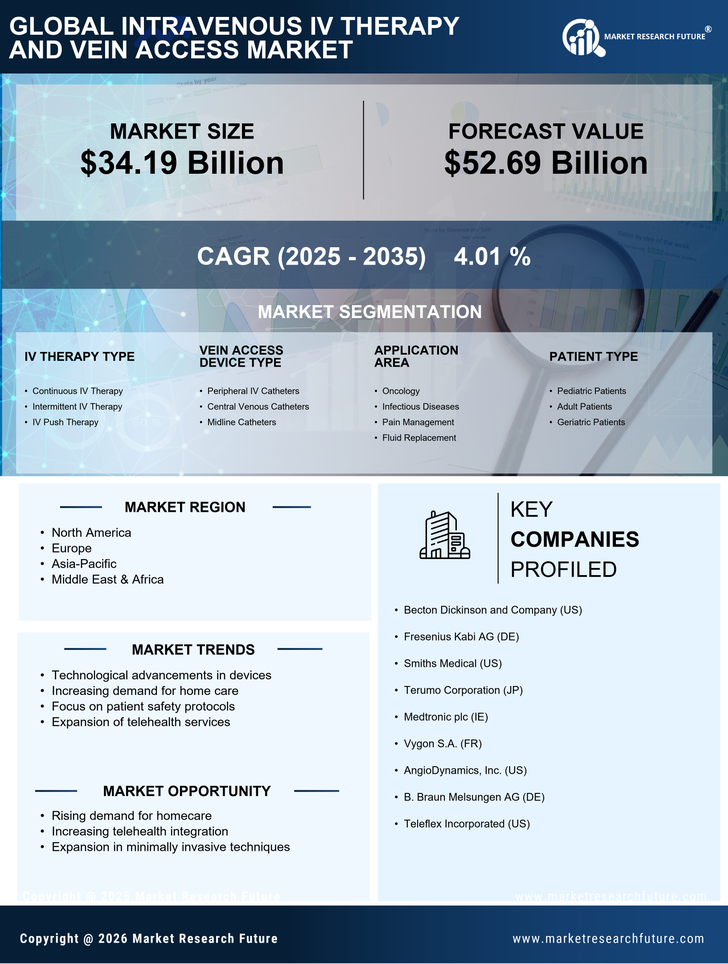
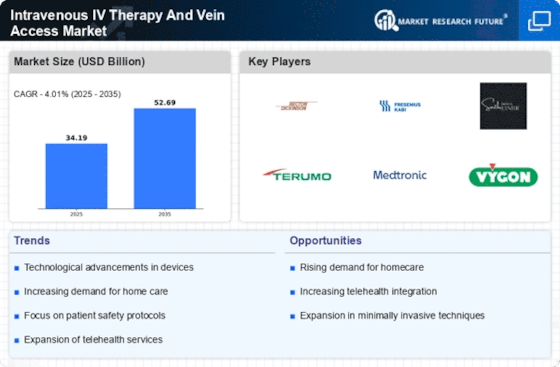
Healthcare Market Segmentation
Healthcare By IV Therapy Type (USD Billion, 2025-2035)
- Continuous IV Therapy
- Intermittent IV Therapy
- IV Push Therapy
Healthcare By Vein Access Device Type (USD Billion, 2025-2035)
- Peripheral IV Catheters
- Central Venous Catheters
- Midline Catheters
Healthcare By Application Area (USD Billion, 2025-2035)
- Oncology
- Infectious Diseases
- Pain Management
- Fluid Replacement
Healthcare By Patient Type (USD Billion, 2025-2035)
- Pediatric Patients
- Adult Patients
- Geriatric Patients
Healthcare By Administration Route (USD Billion, 2025-2035)
- Intravenous
- Intraosseous