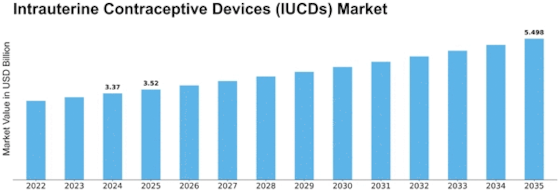
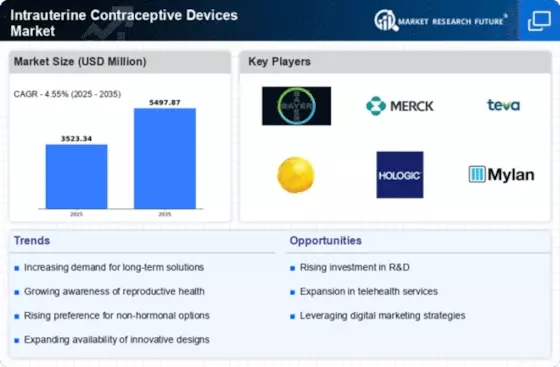
Intrauterine Contraceptive Devices Size
Intrauterine Contraceptive Devices Market Growth Projections and Opportunities
By 2032, the intrauterine contraceptive device (IUCD) market would reach USD 5.71 billion, rising 11.2% annually. The Intrauterine Contraceptive Device Marketing is affected by many factors. All of these factors form and build the industry. People are becoming more aware of and receptive to LARC choices, including IUCDs. IUCD sales have increased as more individuals seek simple, effective family planning methods. IUCDs function well, are simple to use, and last long, supporting this trend. These traits attract safe birth control seekers. Regulatory issues affect the IUCDs market because they ensure safe, effective, and proper usage. IUCDs must be authorized, manufactured, and distributed as prescribed. These standards promote safe business operations and user wellness. Following these criteria ensures IUCDs function and fosters confidence between healthcare staff and users. New contraceptive research and development technologies are transforming the IUCDs industry. IUCD design, materials, and injection processes are constantly updated to enhance user experience, reduce side effects, and increase choice. These technological developments aim to improve user experience, suit patient demands, and encourage more individuals to use IUCDs for birth control. Healthcare finances, long-term contraceptive cost-effectiveness, and inexpensive birth control impact IUCD adoption. IUCDs cost more upfront than some other birth control methods, but they're cheaper over time, offer more advantages, and don't need to be updated as frequently. This attracts healthcare personnel and long-term family planning users. Market dynamics and economic variables determine how simple it is to obtain new IUCD technology. There are several pharmaceutical and medical device businesses that develop various IUCDs, making this industry competitive. Due to competition, companies are developing IUCDs with superior design, less side effects, and personalized options to fit the demands of a broad spectrum of consumers. Healthcare providers may recommend IUCDs for each patient since there are so many alternatives. This helps the market adapt. Demographics including age, sexual desires, and long-term birth control impact market patterns. IUCDs are growing more popular as people realize how they may aid all ages, especially children. People also seek long-term family planning. Population changes alter the requirement for particular IUCDs, which affects product development. Different cultures' views on family planning, birth control, and reproductive health impact the IUCDs industry. Acceptance of long-term birth control, cultural family planning regulations, and knowledge of birth control choices might influence a patient's decision to choose IUCDs. Cultural influences affect healthcare staff' and patients' communication, which impacts IUCD usage choices. Different healthcare facilities, family planning programs, and cultural attitudes impact market considerations. Emerging and established countries may have varied IUCD and family planning information. These gaps are being reduced by improving healthcare systems, educating individuals about birth control options, and removing cultural obstacles. This impacts the worldwide IUCDs market.



















Leave a Comment