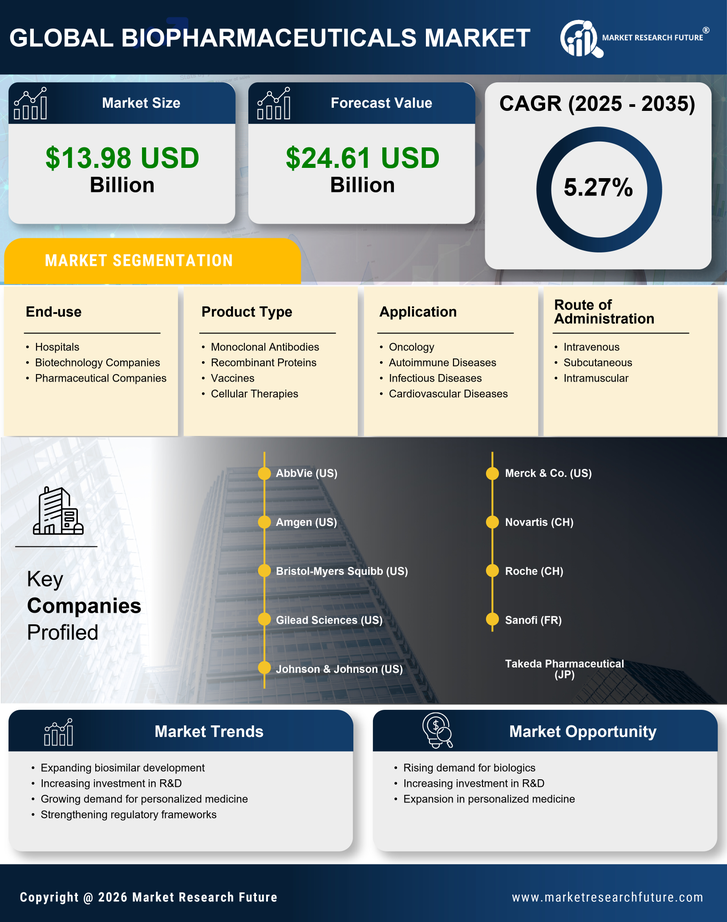
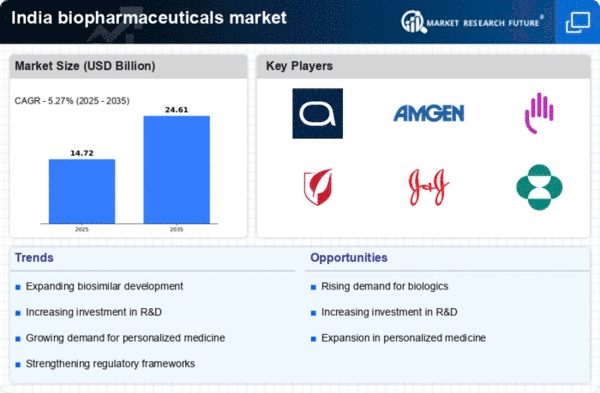
India Construction Market Segmentation
Construction By Product Type (USD Billion, 2025-2035)
- Monoclonal Antibodies
- Recombinant Proteins
- Vaccines
- Cellular Therapies
Construction By Application (USD Billion, 2025-2035)
- Oncology
- Autoimmune Diseases
- Infectious Diseases
- Cardiovascular Diseases
Construction By End-use (USD Billion, 2025-2035)
- Hospitals
- Biotechnology Companies
- Pharmaceutical Companies
Construction By Route of Administration (USD Billion, 2025-2035)
- Intravenous
- Subcutaneous
- Intramuscular