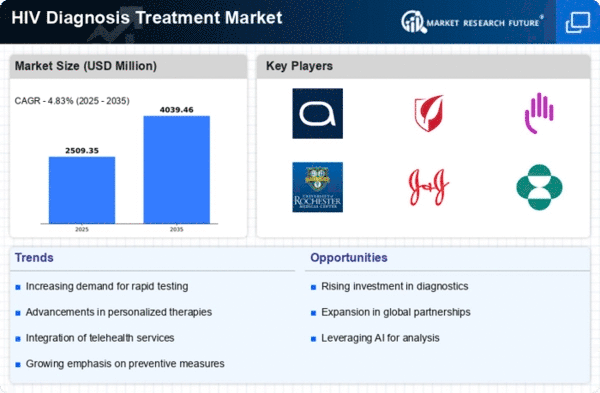
Market Analysis
In-depth Analysis of HIV Diagnosis Treatment Market Industry Landscape
It is a complicated market that reflects the dynamics of HIV diagnosis and treatment, and these have various factors at play in shaping the landscape of this critical healthcare domain. The ongoing prevalence of global HIV/AIDS is one of the main drivers for market expansion necessitating robust diagnostic and treatment options. This has led to advancements in diagnostic technologies due to high demand for accurate and timely diagnosis, leading to a wide variety of testing methods such as nucleic acid tests, rapid tests, enzyme-linked immunosorbent assays (ELISAs), among others. Such diversity is appealing to different kinds of health care settings, contributing to the growth of markets.
Government initiatives and funding are dominant market dynamics. There has been an increase in public health programs all over the world aimed at fighting against HIV infections through testing widely as well as providing timely treatments. Research and development is boosted by government and non-governmental organizations’ support while improving accessibility to diagnostics and treatment options especially in resource constrained areas. In addition, awareness campaigns targeting education also influence market dynamics toward more proactive approach regarding HIV diagnoses together with treatment.
The pharmaceutical industry plays a crucial role in shaping ARTs or antiretroviral therapies that can be used by patients suffering from HIV so that their effectiveness can be improved without causing negative effects on those who use them. The introduction of new drugs into the market emphasizes enhanced patient adherence with improved outcomes in formulating new medications for better results. Partnerships between pharmaceutical companies, research institutions, healthcare organizations dominate the competitive landscape for addressing multiple challenges faced by HIV/AIDS patients.
Market dynamics in recent years have been significantly influenced by technological advances made towards HIV diagnosis and treatment.The landscape changed when point-of-care testing devices came up making decentralization possible thereby allowing quick tests.The integration artificial intelligence and machine learning into diagnostic tools makes them even more efficient while keeping their accuracy intact.
This has necessitated globalization which means sharing best practices and having standardized guidelines across regions. International partnerships and collaborations help in harmonizing diagnostic and treatment approaches hence ensuring uniformity of care across various regions. The interconnectedness also supports the flow of information and resources that makes it possible for all-inclusive efforts to deal with HIV epidemic.
Low-income countries are experiencing disparities in healthcare infrastructure that present market challenges. There is limited access to diagnostic facilities and treatment options, which further indicates why specific interventions must be implemented plus global health initiatives. Apart from this, testing rates by HIV/AIDS victims are kept low due to social stigmas surrounding the disease thereby impeding universal diagnosis as well as provisions of care.



















Leave a Comment