Germany Mitochondrial Myopathy Diagnosis Treatment Market
ID: MRFR/HC/55215-HCR
200 Pages
Satyendra Maurya
Last Updated: April 02, 2026
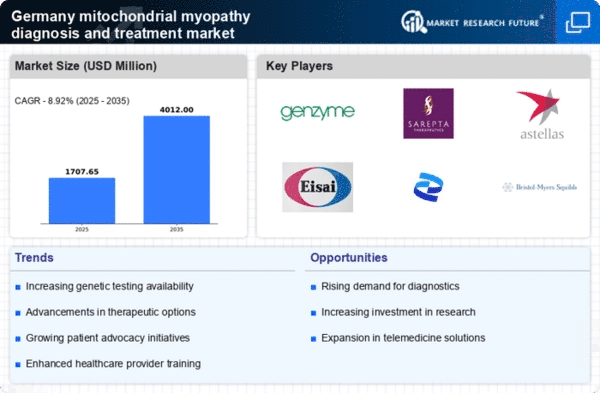
Germany Mitochondrial Myopathy Diagnosis and Treatment Market Research Report: Size, Share, Trend Analysis By Types (Mitochondrial Encephalomyopathy, Lactic Acidosis, and Stroke-Like Episodes, Progressive External Ophthalmoplegia, Myoclonic Epilepsy with Ragged Red Fibers, Mitochondrial Neurogastrointestinal Encephalopathy Syndrome, Kearns–Sayre Syndrome, Mitochondrial DNA Depletion Syndrome, Pearson Syndrome, Leigh Syndrome, Neuropathy, Ataxia, others), By Diagnostic Test (Genetic Tests, Muscle Biopsy, Biochemical Test), By Deployment Models (On-Premise, Cloud-Based) and By Therapy (Supportive Therapy, Targeted Therapy) - Growth Outlook & Industry Forecast 2025 To 2035