Germany Security, Access Control and Robotics Market Segmentation
Security, Access Control and Robotics By Type (USD Million, 2025-2035)
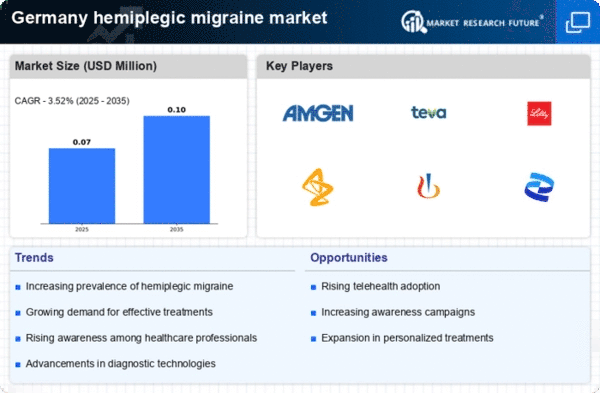
- Familial Hemiplegic Migraine
- Sporadic Hemiplegic Migraine
Security, Access Control and Robotics By Treatment (USD Million, 2025-2035)
- Acute treatment
- Triptans
- Ergotamines
- NSAIDs
- Antiemetics
- Narcotic Analgesics
- Others
Security, Access Control and Robotics By End-user (USD Million, 2025-2035)
- Hospitals & Clinics
- Diagnostic Centers
- Others
Security, Access Control and Robotics By Preventive Treatment (USD Million, 2025-2035)
- Antidepressants
- Botox Injections
- Others
Security, Access Control and Robotics By Distribution Channel (USD Million, 2025-2035)
- Retail Pharmacies
- Hospital Pharmacies
- Online Pharmacies
- Others