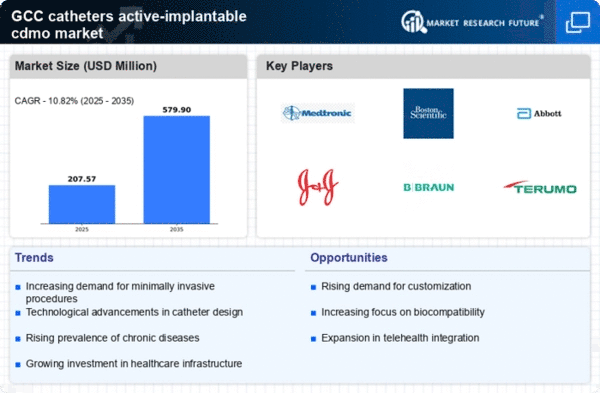
GCC Life Sciences Market Segmentation
Life Sciences By Service Segment (USD Million, 2025-2035)
- Catheters
- Active Implantable Devices
- Endoscopy Devices
Life Sciences By End User (USD Million, 2025-2035)
- Medical Device OEM Companies
- Academic and Research Institutes
- Government Agencies