France Dental Anesthesia Market
ID: MRFR/HC/42990-HCR
200 Pages
Rahul Gotadki
Last Updated: April 24, 2026
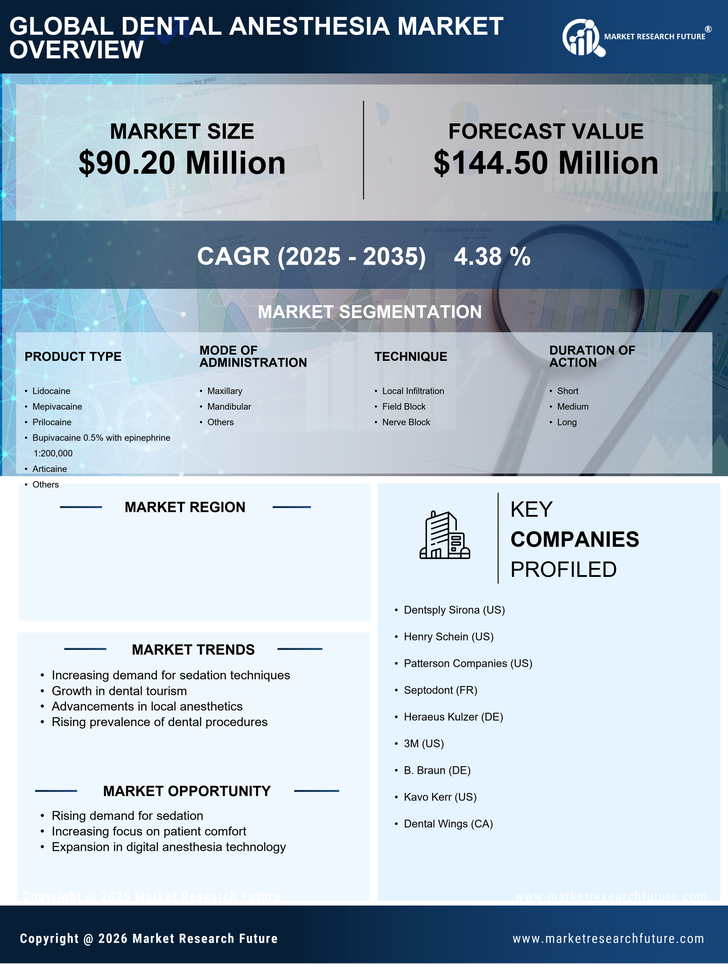
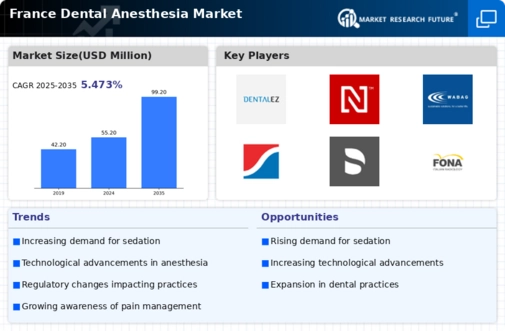
France Dental Anesthesia Market Research Report By Product Type (lidocaine, mepivacaine, prilocaine, bupivacaine 0.5% with epinephrine1:200,000, articaine, others), By Mode of Administration (maxillary, mandibular, others), By Technique (local infiltration, field block, nerve block), By Duration of Action (short, medium, long), By End User (hospital clinics, clinics, other), By Distribution Channel (pharmacies and drug stores, e-commerce, other) and By Intended Audience (hospitals, dental clinics, research institutes, dental software manufacturers, healthcare it companies) - Growth & Industry Forecast 2025 To 2035