North America : Market Leader in Fabry Treatments
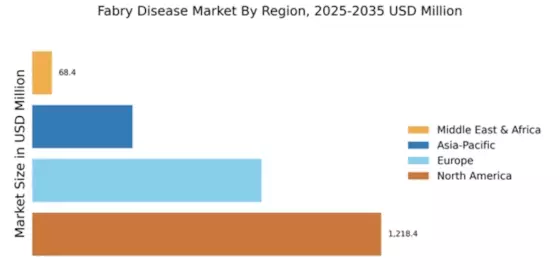
North America leads in the Fabry Disease Market size, accounting for over 50.00% of the global revenue in 2024. The region benefits from advanced healthcare infrastructure, increasing awareness of rare diseases, and supportive regulatory frameworks that encourage innovation. The demand for effective therapies is driven by a growing patient population and the introduction of novel treatments, which are expected to enhance patient outcomes.
The competitive landscape in North America is robust, featuring key players such as Sanofi, Amicus Therapeutics, and Pfizer. These companies are actively engaged in research and development to bring forth innovative therapies. The U.S. remains the largest market, supported by favorable reimbursement policies and a strong focus on patient-centric care. The presence of leading pharmaceutical firms ensures a continuous pipeline of new treatments, further solidifying North America's market position.
- The Centers for Disease Control and Prevention reports that rare diseases affect 25–30 million people in the U.S., creating substantial demand for specialized treatments. Strong healthcare infrastructure and reimbursement systems significantly support adoption of advanced Fabry disease therapies.
Europe : Emerging Market with Growth Potential
Europe Fabry Disease Market size was valued at USD 800 million in 2024, making it the second-largest regional market with a 32.83% share. The region's growth is fueled by increasing awareness among healthcare professionals and patients, alongside supportive regulatory initiatives aimed at improving access to treatments. The European Medicines Agency (EMA) has been instrumental in expediting the approval of innovative therapies, which is expected to drive market expansion in the coming years.
Leading countries in Europe include Germany, France, and the UK, where significant investments in healthcare infrastructure are being made. Key players such as Bayer and Sobi are actively involved in the market, focusing on developing effective therapies. The competitive landscape is characterized by collaborations and partnerships aimed at enhancing treatment options for patients, positioning Europe as a vital player in The Fabry Disease.
- The European Organisation for Rare Diseases indicates that over 30 million people in Europe live with rare diseases, driving policy focus and funding. This supports improved access to therapies and fuels growth in the Fabry disease treatment market across the region.
Asia-Pacific : Emerging Powerhouse in Rare Diseases
The Asia-Pacific region is emerging as a significant player in the Fabry Disease market, with a market size of $350 million in 2024. The growth is driven by increasing healthcare expenditure, rising awareness of rare diseases, and the introduction of new therapies. Governments in countries like Japan and Australia are implementing policies to improve access to treatments, which is expected to further stimulate market growth in the region.
Japan and Australia are leading markets within Asia-Pacific, with key players such as Takeda and Amicus Therapeutics actively participating. The competitive landscape is evolving, with a focus on research and development to address unmet medical needs. As the region continues to invest in healthcare infrastructure, the Fabry Disease market is anticipated to expand significantly, providing new opportunities for stakeholders.
Middle East and Africa : Untapped Market with Challenges
The Middle East and Africa region represents an untapped market for Fabry Disease treatments, with a market size of $68.37 million in 2024. The growth potential is hindered by limited awareness of rare diseases and inadequate healthcare infrastructure. However, increasing investments in healthcare and initiatives to improve disease awareness are expected to drive market growth in the coming years. Regulatory bodies are beginning to recognize the importance of addressing rare diseases, which may lead to improved access to treatments.
Countries like South Africa and the UAE are showing promise in developing their healthcare systems to better address rare diseases. The presence of key players is limited, but companies like Bayer are exploring opportunities in this region. As awareness and infrastructure improve, the competitive landscape is likely to evolve, paving the way for new entrants and innovative therapies.