Europe Welding Fume Extraction Equipment Market
ID: MRFR/Equip/57631-CR
121 Pages
Snehal Singh
Last Updated: January 13, 2026
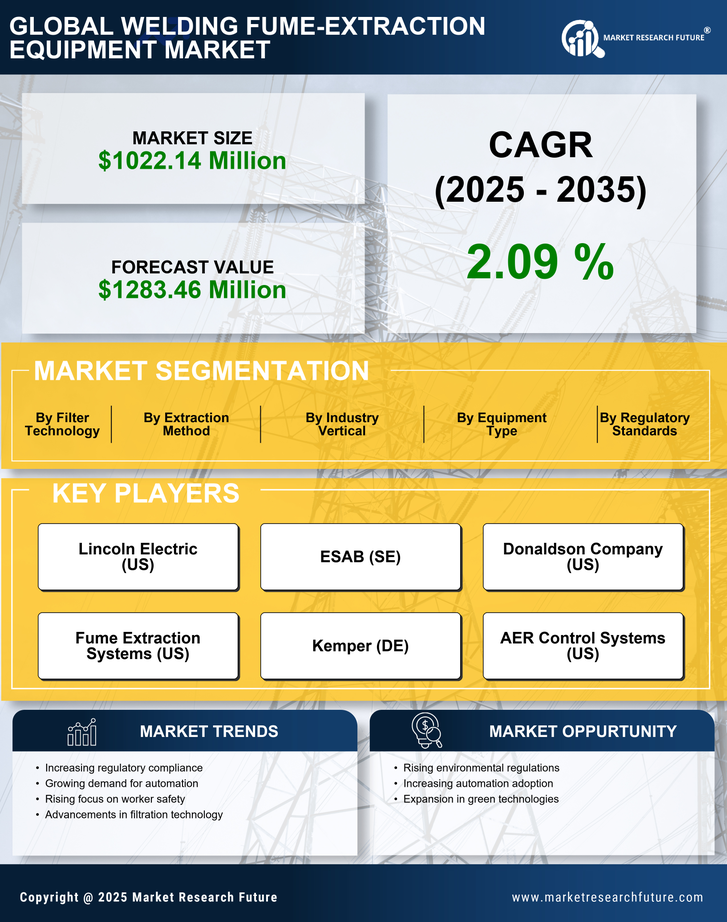
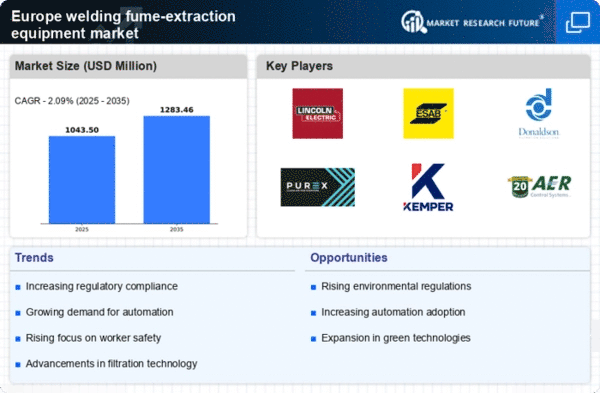
Europe Welding Fume Extraction Equipment Market Research Report Market Research Report Information By Product type (Downdraft Fume Extractors, Centralized Fume Extraction Systems, On-Torch Fume Extraction Systems, and Others), By Technology (High Vacuum Extraction, Low Vacuum Extraction, Electrostatic Precipitation, Filtration/HEPA Filters, Activated Carbon Filters, and Others), By Application(Metal Welding, Plastic Welding, and Others) By End-User Industry(Automotive, Aerospace & Defense, Shipbuilding, Manufacturing & Fabrication, Construction, Oil & Gas, Heavy Equipment Manufacturing, Electronics & Semiconductors, and Others ) By extraction method, By power rating and by Country –Market Forecast Till 2035