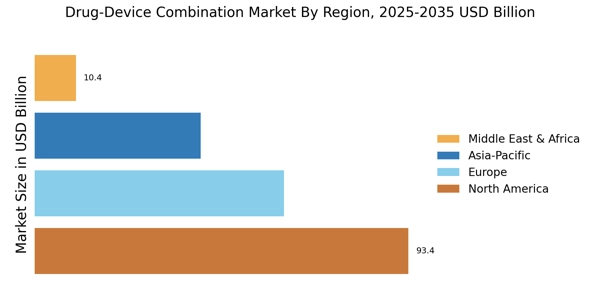
Regulatory Support
The Drug-Device Combination Market benefits from evolving regulatory frameworks that aim to streamline the approval processes for innovative products. Regulatory bodies are increasingly recognizing the potential of drug-device combinations to address unmet medical needs, leading to more favorable guidelines and expedited review pathways. For instance, the FDA has introduced programs such as the Breakthrough Devices Program, which aims to accelerate the development of devices that provide more effective treatment options. This regulatory support is crucial, as it not only encourages investment in research and development but also enhances market access for new products. As a result, the Drug-Device Combination Market is likely to see a rise in the number of approved products, fostering competition and innovation.
Patient-Centric Approaches
The Drug-Device Combination Market is witnessing a shift towards patient-centric approaches that prioritize user experience and outcomes. Manufacturers are increasingly focusing on designing devices that are not only effective but also easy to use, thereby enhancing patient compliance. This trend is evident in the development of user-friendly interfaces and mobile applications that facilitate better interaction between patients and their treatment regimens. Research indicates that patient adherence to prescribed therapies can significantly improve health outcomes, which in turn drives demand for innovative drug-device combinations. As healthcare systems continue to emphasize patient engagement, the Drug-Device Combination Market is likely to evolve, with a greater emphasis on personalized medicine and tailored treatment solutions.
Technological Advancements
The Drug-Device Combination Market is experiencing a surge in technological advancements that enhance the efficacy and safety of drug delivery systems. Innovations such as smart inhalers and implantable devices are becoming increasingly prevalent, allowing for more precise dosing and improved patient adherence. According to recent data, the market for smart drug delivery devices is projected to reach approximately 20 billion USD by 2026, indicating a robust growth trajectory. These advancements not only improve therapeutic outcomes but also facilitate real-time monitoring of patient health, thereby fostering a more proactive approach to treatment. As technology continues to evolve, the Drug-Device Combination Market is likely to witness further integration of artificial intelligence and machine learning, which could revolutionize patient care and streamline drug development processes.
Increased Investment in R&D
The Drug-Device Combination Market is experiencing a notable increase in investment in research and development, driven by the need for innovative solutions to complex health challenges. Pharmaceutical and medical device companies are allocating substantial resources to develop new drug-device combinations that can address specific therapeutic needs. This trend is supported by various funding initiatives and partnerships between industry stakeholders, academic institutions, and government agencies. For instance, recent reports indicate that investment in medical technology R&D has grown by over 10% annually, reflecting a strong commitment to innovation. As a result, the Drug-Device Combination Market is poised for growth, with a pipeline of new products that could transform patient care and treatment outcomes.
Rising Prevalence of Chronic Diseases
The Drug-Device Combination Market is significantly influenced by the rising prevalence of chronic diseases, which necessitate advanced treatment options. Conditions such as diabetes, cardiovascular diseases, and respiratory disorders are becoming increasingly common, driving demand for effective drug delivery systems. For example, the World Health Organization reports that chronic diseases account for approximately 71% of all deaths globally, underscoring the urgent need for innovative solutions. Drug-device combinations, such as insulin pumps and inhalers, are particularly well-suited to manage these conditions, offering patients improved quality of life and better disease management. This growing demand is expected to propel the Drug-Device Combination Market forward, as healthcare providers seek to implement more effective treatment strategies.