Market Analysis
In-depth Analysis of Clinical Trials Market Industry Landscape
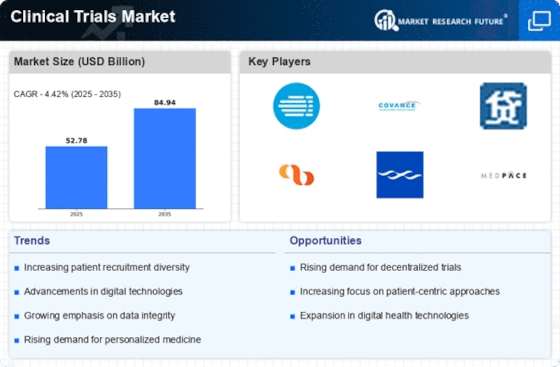
Within the healthcare and pharmaceutical industries, the clinical trials market is distinct and plays a critical role in the advancement of novel drugs, treatments, and therapeutic interventions. The growing global need for clinical investigation is driving the market's components. The need for clinical studies to evaluate the safety and efficacy of novel medicines is growing as the global burden of infections rises. This requirement is particularly clear in developing countries where the framework for medical services is expanding. Wearable technology, information analysis, and electronic data collection contribute to more successful and patient-driven clinical studies. These developments improve the accuracy of the information, shorten the preliminary period, and advance the overall preliminary findings. Understanding the factors considered during the initial planning phase, fostering dedication, and ensuring accurate depiction are becoming increasingly important. The general outcome, retention, and enrolment of clinical studies are all improved by this patient-driven methodology. The intricacy of clinical preliminary conventions is on the ascent, affected by the need to address assorted patient populaces, complex illnesses, and customized medication. This unique shift requires versatile preliminary plans and adaptable approaches to oblige developing logical information and administrative necessities. Clinical trials are progressively globalized, with supports directing trials in various nations to get to assorted patient populaces and facilitate enrolment. The globalization pattern is impacted by the requirement for more agent preliminary information, proficient enlistment, and cost-adequacy in preliminary direct. CROs assume a critical part in leading trials, offering skill, framework, and versatility. Coordinated efforts among supporters and CROs are fundamental for streamlining preliminary productivity and guaranteeing ideal consummation. Certifiable Proof (RWE) is acquiring conspicuousness in the clinical trials market. The incorporation of RWE close by customary clinical preliminary information gives a more complete comprehension of treatment results and supports administrative direction. The joining of RWE adds to the unique idea of preliminary plan and investigation. Drug companies and payers are beginning to see some progress with their esteem-based agreements, which link payments to treatment outcomes. This change necessitates innovative first strategies that demonstrate the value and suitability of novel therapies. The market for clinical trials is being formed in large part by public-private partnerships. These groups tackle issues with medical services more broadly and expedite the development of new medications.



















Leave a Comment