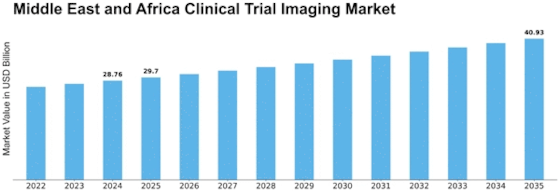
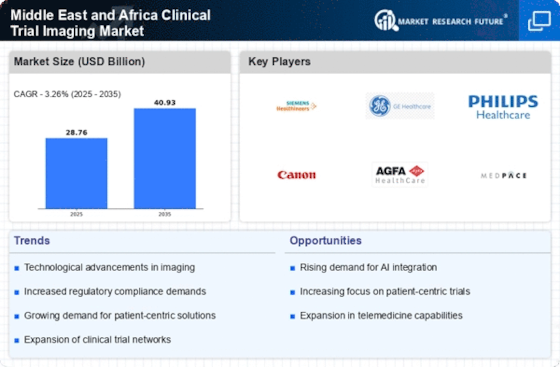
Clinical Trial Imaging Size
Clinical Trial Imaging Market Growth Projections and Opportunities
Increasing clinical studies drive the Middle East and Africa clinical trial imaging market. In the Middle East and Africa, more industrial and university institutions are doing clinical studies, which needs the most up-to-date imaging tools. The pharmaceutical and biotechnology businesses in the Middle East and Africa are driving demand for clinical study images. Clinical trial imaging is greatly enhanced by this crucial component. Due to the increase and investment in these areas, cutting-edge imaging technologies are needed to efficiently explore and develop new medications. Government measures to increase healthcare research and development affect the clinical trial imaging industry. These initiatives increase clinical trial imaging demand. Governments in the Middle East and Africa fund and cooperate to improve clinical trial infrastructure, making imaging technology adoption possible. Imaging Contract Research Organizations (CROs) in the Middle East and Africa are a key market trend. These firms specialize in clinical imaging services. Since their expertise and technology are tailored to pharmaceutical and biotechnology research in the area, they may profit from it. MRI, CT, and PET technology advancements are driving the clinical trial imaging industry. Imaging advances have improved clinical trial evaluations, encouraging sponsors and researchers to participate. International cooperation in clinical research is affecting the market for clinical trial imaging. Imaging systems that meet global standards and integrate data are needed more. Collaborations between international pharmaceutical companies, research institutes, and African and Middle Eastern organizations are driving this product's market. The diverse Middle East and Africa patient population is a key market factor. Clinical studies often include many participants to guarantee generalizability. Due to the region's diverse population, imaging technologies that can meet the demands of several patient groups are needed. Clinical studies using cutting-edge imaging technology are subject to strict regulatory compliance. Modern imaging technologies are needed to meet worldwide regulatory authorities' data accuracy and safety criteria. This pushes the demand for suitable, cutting-edge imaging equipment for clinical studies. The clinical trial imaging market reflects the healthcare industry's expanding focus on precision medicine. Imaging technologies are crucial to customizing treatment techniques. Precision medicine is growing across the Middle East and Africa, necessitating better clinical trial imaging. Healthcare infrastructure growth is a crucial market factor. Modern medical facilities with cutting-edge imaging technology are making the Middle East and Africa more appealing for clinical trials. Thus, imaging services in advanced healthcare facilities are in high demand.



















Leave a Comment