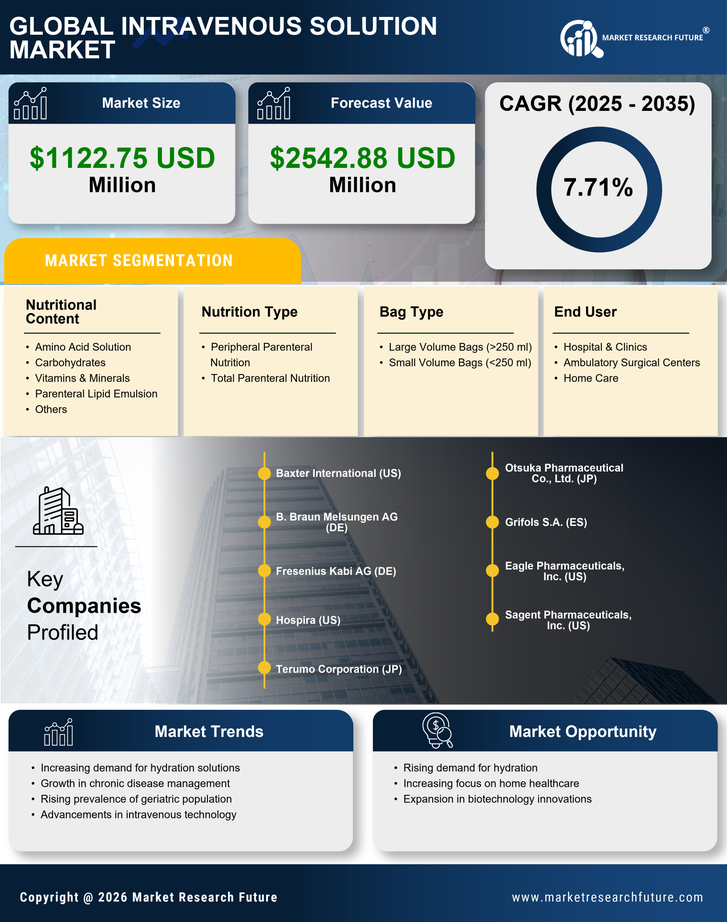
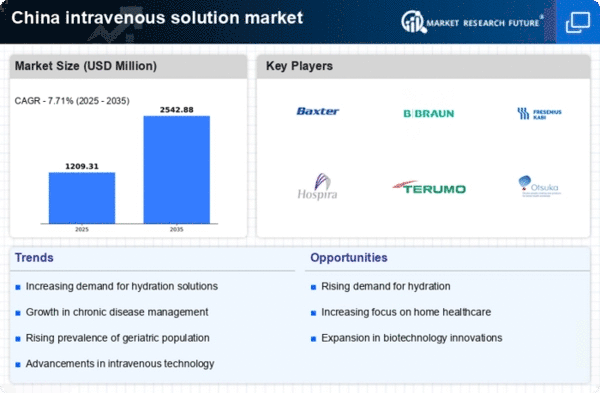
China Medical Device Market Segmentation
Medical Device By Nutrition Type (USD Million, 2025-2035)
- Peripheral Parenteral Nutrition
- Total Parenteral Nutrition
Medical Device By Bag Type (USD Million, 2025-2035)
- Large Volume Bags (>250 ml)
- Small Volume Bags (<250 ml)
Medical Device By Nutritional Content (USD Million, 2025-2035)
- Amino Acid Solution
- Carbohydrates
- Vitamins & Minerals
- Parenteral Lipid Emulsion
- Others
Medical Device By End User (USD Million, 2025-2035)
- Hospital & Clinics
- Ambulatory Surgical Centers
- Home Care