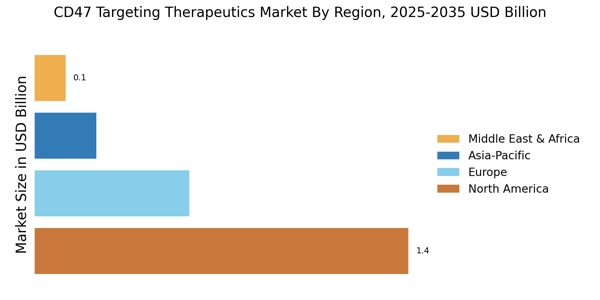
The CD47 Targeting Therapeutics Market is currently experiencing a notable evolution, driven by advancements in immunotherapy and a growing understanding of the role of CD47 in cancer biology. This market appears to be gaining traction as researchers and pharmaceutical companies explore innovative approaches to harness the immune system's potential against tumors. The increasing prevalence of various cancers, coupled with the limitations of traditional therapies, suggests a shift towards targeted treatments that can enhance patient outcomes.
Furthermore, collaborations between academic institutions and biotech firms seem to be fostering a rich environment for the development of novel CD47-targeting agents, which may lead to breakthroughs in treatment options. In addition, regulatory bodies are likely to play a crucial role in shaping the landscape of the market. As more clinical trials progress, the approval of new therapies could pave the way for expanded access to these innovative treatments.
The market's future appears promising, with ongoing research and development efforts indicating a potential for significant growth. Stakeholders are increasingly recognizing the importance of personalized medicine, which may further drive interest in CD47-targeting strategies. Overall, the market is poised for transformation, with the potential to redefine cancer treatment paradigms.
Rising Interest in Immunotherapy
The CD47 Targeting Therapeutics Market is witnessing a surge in interest surrounding immunotherapy. This trend reflects a broader shift towards harnessing the body's immune system to combat cancer. As more studies highlight the efficacy of CD47-targeting agents, stakeholders are increasingly investing in research and development to explore their potential.
Research activity around CD47 inhibitors is accelerating, with more than 25 clinical trials globally evaluating CD47-targeting antibodies and combination therapies for cancer treatment, highlighting the growing interest in immune checkpoint modulation.
- The World Health Organization (WHO) reports approximately 10 million cancer-related deaths annually, while the Centers for Disease Control and Prevention (CDC) highlights over 1.9 million new cancer cases each year in the U.S., reinforcing strong global demand for immunotherapy approaches, including CD47-targeting therapies improving survival outcomes.
Collaborative Research Initiatives
Collaborations between academic institutions and biotechnology companies are becoming more prevalent in the CD47 Targeting Therapeutics Market. These partnerships aim to accelerate the discovery and development of novel therapies. By pooling resources and expertise, stakeholders are likely to enhance the pace of innovation and bring new treatments to market.
Immunotherapy continues to dominate oncology innovation, with over 40% of oncology drugs currently in the global clinical pipeline focusing on immunotherapy approaches, including checkpoint inhibitors such as CD47-targeting therapies.
- Gavi, the Vaccine Alliance has supported immunization of over 1 billion children through global partnerships, while Médecins Sans Frontières (MSF) operates in more than 70 countries delivering collaborative healthcare programs, highlighting the effectiveness of partnerships in accelerating research innovation and therapeutic development pipelines globally.
Regulatory Advancements
Regulatory bodies are adapting to the evolving landscape of the CD47 Targeting Therapeutics Market. As clinical trials yield promising results, there appears to be a growing willingness to expedite the approval process for innovative therapies. This trend may facilitate quicker access to groundbreaking treatments for patients, potentially transforming standard care practices.
Regulatory agencies such as the U.S. Food and Drug Administration are accelerating oncology innovation, with over 60% of new cancer drug approvals in recent years receiving priority review or expedited regulatory pathways, supporting the development of novel targeted treatments.
- The Pan American Health Organization (PAHO) supports regulatory strengthening across over 30 countries in the Americas, while the European Centre for Disease Prevention and Control (ECDC) coordinates health surveillance across 30 EU/EEA nations, indicating increasing regulatory alignment that supports faster approval pathways for innovative therapies.