Cardiopulmonary Resuscitation Market
ID: MRFR/MED/0840-CR
154 Pages
Nidhi Mandole
Last Updated: May 11, 2026
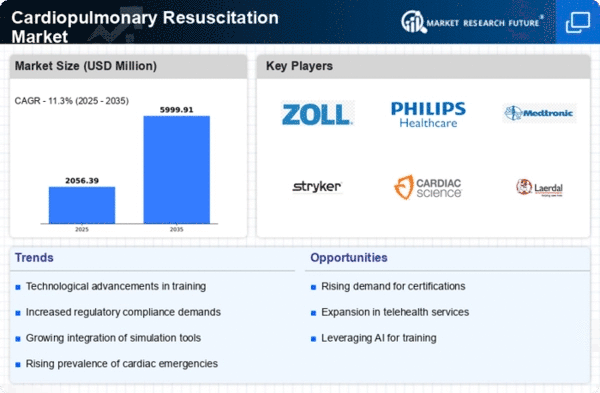
Cardiopulmonary Resuscitation Market Research Report Information By Product Type (Piston CPR Devices, Load- Disturbing Band (LBD), Active Compression and Impedance Threshold Device (ACD+ITD), Extracorporeal Techniques and Invasive Perfusion Devices, and Others), By Compressor Type (Automatic Mechanical (Battery Driven, Pneumatic Driven and Mechanically Actuated and Battery Driven) and Manual CPR), By Application (EMT Rescue Units, Coronary & Intensive Care Units, Emergency Departments, Organ Transplants Facilities, Air Medevac Units, and Others), By End User (Hospitals & Clinics, Cardiac Catheterization Laboratories, Organ Transplant Unit, and Others), And By Region (North America, Europe, Asia-Pacific, And Rest Of The World) –Market Forecast Till 2035