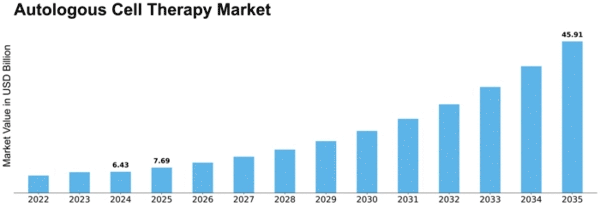
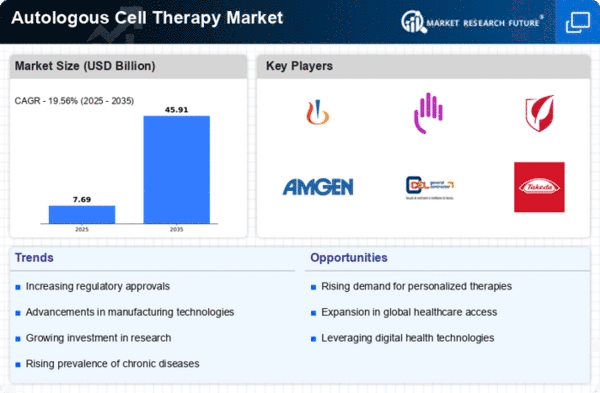
Autologous Cell Therapy Size
Autologous Cell Therapy Market Growth Projections and Opportunities
Among factors driving the autologous cell therapy market is a range of them influencing its growth and developments. One of primary forces that promote autologous cell therapy is the greater contribution invested in therapeutic potential of autologous cell treatment for treating different medical states. Several initiatives have emerged, making the concept of autologous cells use for treatment purposes popular and contributing to the increasing revival in calls for individualized and regenerative treatment options. Technological developments have a crucial role to effect on the autologous cell therapy market. The growth of further cell therapies is stemmed from the ever-evolving practices that may come to pass in cell isolation, expansion, and manipulation, including advancements technology for gene editing such as CRISPR-Cas9. This technological innovation not only enhances the performance of autologous cell therapies but also broadens their scope in terms of widening the diseases and conditions that they are to be used on. Shaping the market dynamics by cell therapy regulations is among the important aspects of cell therapies. The security, efficacy and quality of autologous cell therapies is assured by strict regulations which will be influence their approval the way in which they are commercialized and placed in clinical settings. These rules should be followed for establishing trust and market entry as trusted institution with healthcare providers, patients, and regulatory bodies. As for autologous cell therapies, the adoption rate varies in different regions due to a range of economic factors and healthcare delivery systems. Factors like the degree of healthcare utilization, reimbursement policies as well as economic status result in affecting the level at which cell therapy is used and patient demand for personalized regenerative treatments. It means that the players are operating in different landscapes and failing to produce products addressing specific challenges posed by these difference healthcare systems. The competitive situation in the market of autologous cell therapy is characterized by a constant process of innovation and development. Currently, biotechnology companies and research institutions focus on the wide range of cell therapies that could be presented with certain features available next to them such as safety and efficiency that is demonstrated in targeting specific diseases. In a competitive environment, the market players continuously face pressure to maintain leading positions in terms of innovation; such an innovative impulse facilitates bringing new technologies to the field of autologous therapies and constantly improving their properties. There are initiatives between cell therapy developers, research institutions as well as healthcare organizations that help in developing innovations in the global tissue and biopsy specimen processing market. The partnerships discussed above concentrate around initiatives like the development new cell sources, clearance of manufacturing process and advances in PMEA approaches. Cooperation between various stakeholders fiui1deceUs transforms the rate of development in autologous cell therapies that meet the demands of changing patients and healthcare workers.



















Leave a Comment