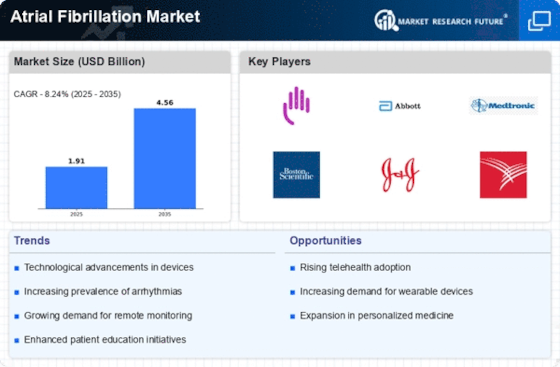
Market Share
Atrial Fibrillation Systems Market Share Analysis
Within the field of cardiovascular health, the Atrial Fibrillation (AF) Market is undergoing distinct processes to determine its market share. Organizations in this field are resolutely investigating the serious scene in order to enhance their market position, with a focus on providing solutions for managing and treating atrial fibrillation. Innovation in improvements for clinical equipment is becoming more and more important to organizations. This includes advancements in embedded devices, removal techniques, and catheter-based chores. Improving atrial fibrillation treatment choices' accuracy, security, and sufficiency is the goal of ongoing development. Businesses are aggressively entering new overseas markets. This include working with providers of medical services, modifying treatment plans to meet various legal requirements, and making sure that cutting edge advancements are available in different regions. Businesses are focusing on non-invasive treatment options in response to patient demands and improvements in non-invasive technologies. This provides patients with additional options for managing their atrial fibrillation, including improved lifestyle choices, medication therapies, and remote monitoring devices. Companies fund educational programs for healthcare workers. Meetings, events, and educational resources provide more details on the most recent choices, suggestions, and innovations in atrial fibrillation treatment. Because atrial fibrillation is common and early treatment is crucial, companies are investigating ways to reduce the cost of this treatment. This involves agreements with healthcare payers, payment techniques, and price models to make atrial fibrillation treatments more accessible for patients. Progress is essential, especially when it comes to telehealth and remote monitoring. Businesses are investing in R&D to develop telehealth platforms and remote monitoring equipment that will enable ongoing patient monitoring, timely intervention, and treatment plan adjustments. Organizations that embrace the cutting edge of medical treatment focus on regular communication using EHR. This ensures that data on atrial fibrillation therapy is linked to patient records, promoting comprehensive medical services across the board and enabling medical care providers to make well-informed decisions. For patient-driven care, organizations are changing their responses to atrial fibrillation. Patients are guaranteed to receive individualized and thorough attention when treatment regimens are tailored based on their preferences, lifestyle factors, and co-occurring conditions. The time for developing research on personalized medicine is almost here. Businesses are investigating genetic and biomarker-based approaches to customize atrial fibrillation therapies for individual patients, aiming to maximize therapeutic benefits and minimize adverse effects. Businesses are considering integrating artificial intelligence (AI) into treatment decision-making. Artificial intelligence algorithms analyze patient data to assist healthcare providers in making more informed decisions regarding the best atrial fibrillation treatment methods.


















Leave a Comment