North America : Market Leader in Animal Vaccines
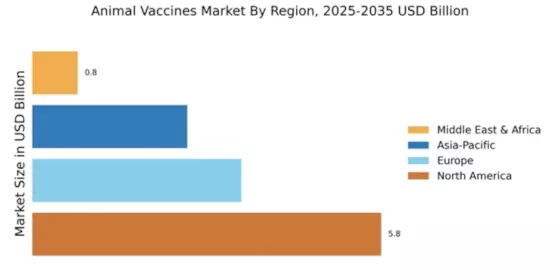
North America continues to lead The Animal Vaccines, holding a significant share of 5.76 in 2024. The region's growth is driven by increasing pet ownership, rising livestock production, and stringent regulations promoting animal health. The demand for innovative vaccines is further fueled by advancements in biotechnology and a growing awareness of zoonotic diseases. Regulatory bodies are actively supporting vaccine development, ensuring safety and efficacy, which enhances market growth.
The competitive landscape in North America is robust, featuring key players such as Zoetis, Merck Animal Health, and Elanco Animal Health. The U.S. is the largest market, driven by high investments in R&D and a strong veterinary infrastructure. Canada also contributes significantly, focusing on sustainable animal farming practices. The presence of established companies and a growing number of startups in the biotech sector are expected to further enhance market dynamics.
Europe : Emerging Market with Growth Potential
Europe's animal vaccines market is valued at 3.45, reflecting a growing demand driven by increasing livestock farming and pet ownership. The region benefits from stringent regulations that ensure high safety standards for vaccines, fostering consumer trust. Additionally, the European Union's initiatives to promote animal health and welfare are significant growth catalysts. The Italy animal vaccines market serves as a key contributor to this regional growth, supported by a robust veterinary infrastructure and high standards for disease prevention in both livestock and companion animals.
The rising incidence of animal diseases necessitates effective vaccination strategies, further propelling market expansion. Leading countries in this region include Germany, France, and the UK, where major players like Boehringer Ingelheim and Virbac are prominent. The competitive landscape is characterized by innovation and collaboration among companies to develop advanced vaccine solutions. The presence of regulatory bodies ensures that products meet high standards, which is crucial for market acceptance. This environment encourages investment in R&D, positioning Europe as a key player in The Animal Vaccines.
Asia-Pacific : Rapidly Growing Animal Health Sector
The Asia-Pacific region is rapidly emerging as a significant player in the animal vaccines market, with a market size of 2.56. The growth is driven by increasing livestock production, rising pet ownership, and a growing awareness of animal health. Governments are implementing supportive regulations to enhance animal welfare, which is further propelling the demand for vaccines. The region is also witnessing advancements in veterinary healthcare, contributing to market expansion.
Countries like China, India, and Australia are leading the market, with key players such as Zoetis and Merck Animal Health establishing a strong presence. The competitive landscape is evolving, with local companies also gaining traction. The increasing collaboration between governments and private sectors is expected to enhance vaccine accessibility and affordability, further driving market growth.
Middle East and Africa : Untapped Market with Growth Opportunities
The Middle East and Africa region, with a market size of 0.75, presents untapped opportunities in the animal vaccines market. The growth is driven by increasing livestock farming and a rising awareness of animal health issues. Governments are beginning to implement regulations to improve animal welfare, which is expected to boost vaccine demand. The region's diverse agricultural practices also create a unique landscape for vaccine development tailored to local needs.
Leading countries in this region include South Africa and the UAE, where there is a growing focus on veterinary healthcare. The competitive landscape is characterized by a mix of local and international players, with companies like Ceva Santé Animale and Virbac making significant inroads. The increasing investment in veterinary services and infrastructure is anticipated to enhance market dynamics and accessibility to vaccines.