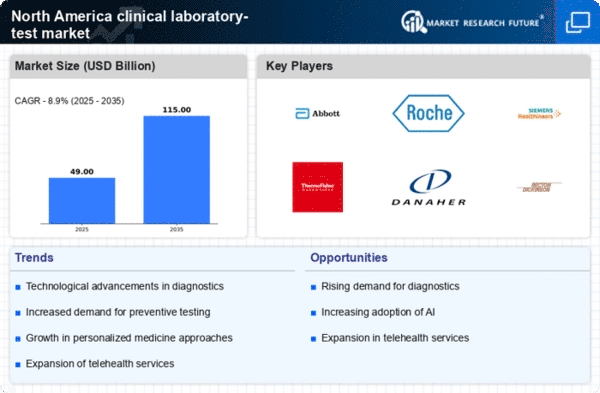
Top Industry Leaders in the North America Clinical Laboratory Test Market

Latest North America Clinical laboratory test Companies Update
-
July 2023: The Atellica CI Analyzer for immunoassay and clinical chemistry testing, the newest addition to Siemens Healthineers' Atellica in vitro diagnostics range, has acquired FDA clearance and is now accessible in many of the world's major markets. Improved turnaround time predictability, sophisticated reporting features, and laser-focused safety and security precautions are just a few of the ways in which the analyzer gives laboratories with low to medium testing volumes a competitive edge. Due to a lack of qualified workers, laboratories of all sizes have significant difficulties in areas such as ensuring the safety of their employees, keeping their data secure, and meeting their environmental objectives, among many others. It might also be used to determine an individual's likelihood of developing Alzheimer's disease and could be of service to pharmaceutical companies in identifying early-stage patients for inclusion in treatment trials.
October 2023: For the detection of gene fusions, translocations, and rearrangements across 361 genes in formalin-fixed, paraffin-embedded (FFPE) tumor tissue, AventaTM Genomics, LLC, a clinical laboratory founded as a joint venture between Arima Genomics, Inc. and Protean BioDiagnostics, Inc., launched Aventa FusionPlus in October 2023. CLIA-accredited testing is carried out in Aventa's Florida lab. The Aventa FusionPlus test uses 3D genomics technology, which takes advantage of the fact that fused and rearranged genes are kept in close proximity to one another in order to increase the signal by a factor of 100 to 1000 and to discover previously unknown breakpoints and fusion partners. The Aventa FusionPlus test found potentially actionable variations in 50% of previously described tumor specimens from patients with no known actionable cause.
- Aurora Diagnostics,
- Laboratory Corporation of America,
- LifeLabs Medical Laboratories,
- Quest Diagnostics