Active Pharmaceutical Ingredients Market Summary
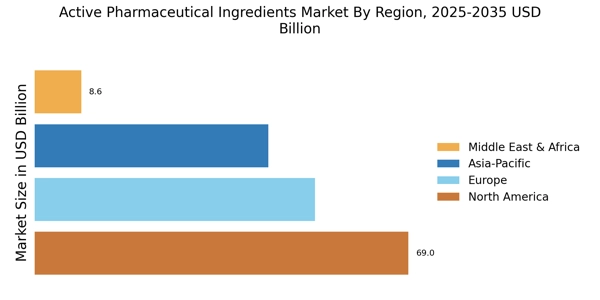
According to MRFR analysis, the Active Pharmaceutical Ingredients Market Size was valued at USD 172.6 Billion in 2024. The market is projected to grow from USD 184.54 Billion in 2025 to USD 360.33 Billion by 2035, registering a CAGR of 6.92% during the forecast 2025–2035. North America led the market with over 39.98% share, generating around USD 69 billion in revenue.
The Active Pharmaceutical Ingredients Market is expanding due to rising global disease burden, increasing demand for generic and specialty medicines, and growth in pharmaceutical manufacturing. Key trends include shift toward complex and high-potency APIs, expansion of outsourcing, and strengthening of global supply chains to ensure consistent drug availability and affordability.
The World Health Organization highlights that noncommunicable diseases account for nearly 74% of global deaths, significantly increasing demand for long-term pharmaceutical treatments and APIs.
Key Market Trends & Highlights
The Active Pharmaceutical Ingredients Market is experiencing robust growth driven by evolving healthcare needs and technological advancements.
- The demand for biologics is surging, reflecting a broader trend towards innovative therapies.
- Quality and compliance remain paramount as regulatory frameworks tighten across the industry.
- Sustainability initiatives are gaining traction, influencing production practices and supply chain management.
- The increasing prevalence of chronic diseases and advancements in pharmaceutical research are key drivers propelling market expansion, particularly in North America and the Asia-Pacific region.
Market Size & Forecast
| 2024 Market Size | 172.6 (USD Billion) |
| 2035 Market Size | 360.33 (USD Billion) |
| CAGR (2025 - 2035) | 6.92% |
Major Players
Companies such as BASF SE (DE), Boehringer Ingelheim GmbH (DE), Teva Pharmaceutical Industries Ltd. (IL), Sandoz International GmbH (CH), Lonza Group AG (CH), Aurobindo Pharma Limited (IN), Fujifilm Diosynth Biotechnologies (JP), Hikma Pharmaceuticals PLC (GB), Mylan N.V. (NL), Dr. Reddy's Laboratories Ltd. (IN) are some of the major participants in the Global Active Pharmaceutical Ingredients Market.