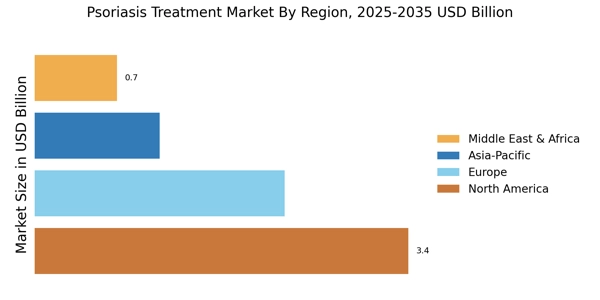Global market valuation was derived through pharmaceutical revenue mapping, prescription volume analysis, and procedure-based phototherapy utilization metrics. The methodology included:
Identification of 40+ key pharmaceutical and medical device manufacturers across North America, Europe, Asia-Pacific, Latin America, and Middle East/Africa, including innovators in monoclonal antibodies (IL-17A, IL-12/23, IL-23p19 inhibitors), biosimilar developers, topical formulation specialists, and phototherapy equipment providers.
Product mapping across biologics (originator and biosimilar TNF inhibitors, IL inhibitors), systemic small molecules (methotrexate, cyclosporine, apremilast, deucravacitinib), topical therapies (high-potency corticosteroids, vitamin D3 analogs, fixed-dose combinations, topical calcineurin inhibitors), and phototherapy devices (narrowband UVB, excimer laser systems).
Analysis of reported and modeled annual revenues specific to psoriasis indications, segmented by treatment line (first-line topicals vs. second-line biologics) and severity (mild, moderate, severe).
Coverage of manufacturers representing 75-80% of global market share in 2024, reflecting the concentrated nature of the biologics market dominated by major immunology players.
Extrapolation using bottom-up (diagnosed prevalent patient population × treatment penetration rates × average treatment cost by country/severity) and top-down (pharmaceutical company reported revenue validation, adjusted for psoriasis-specific indication where available) approaches to derive segment-specific valuations for biologics, biosimilars, and conventional therapies.
Price elasticity modeling for biosimilar entry scenarios and formulary placement impact across hospital, dermatology clinic, and homecare administration settings.