
Artificial Eye Market
Rapport d'étude de marché sur les yeux artificiels par type (implants non intégrés, implants intégrés), technologie (électronique et mécanique), utilisateurs finaux (hôpitaux, cliniques et autres) - Prévisions jusqu'en 2027
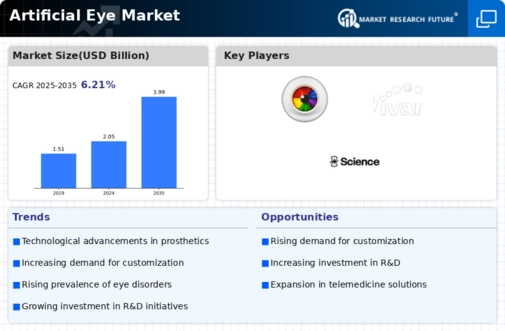
-
1 INTRODUCTION
- 1.1 DEFINITION
-
1.2 SCOPE OF STUDY
- 1.2.1 RESEARCH OBJECTIVE
-
1.2.2 ASSUMPTIONS & LIMITATIONS
- 1.2.2.1 ASSUMPTIONS
- 1.2.2.2 LIMITATIONS
- 1.3 MARKET STRUCTURE:
-
2 RESEARCH METHODOLOGY
- 2.1 RESEARCH PROCESS:
- 2.2 PRIMARY RESEARCH
- 2.3 SECONDARY RESEARCH:
-
3 MARKET DYNAMICS
- 3.1 DRIVERS
- 3.2 RESTRAINTS
- 3.3 OPPORTUNITIES
- 3.4 MACROECONOMIC INDICATORS
-
4 MARKET FACTOR ANALYSIS
- 4.1 PORTERS FIVE FORCES MODEL
- 4.2 BARGAINING POWER OF SUPPLIERS
- 4.3 BARGAINING POWER OF BUYERS
- 4.4 THREAT OF NEW ENTRANTS
- 4.5 THREAT OF SUBSTITUTES
- 4.6 INTENSITY OF RIVALRY
-
5 GLOBAL ARTIFICIAL EYE Artificial Eye Market, BY SURGERY
- 5.1 EVISCERATION SURGERY
- 5.2 ENUCLEATION SURGERY
-
6 GLOBAL ARTIFICIAL EYE Artificial Eye Market, BY MATERIAL TYPE
- 6.1 CRYOLITE GLASS
- 6.2 HARD PLASTIC ACRYLIC
-
7 GLOBAL ARTIFICIAL EYE Artificial Eye Market, BY PRODUCT TYPE
- 7.1 MOULDED
- 7.2 COSMETIC SHELL
-
8 GLOBAL ARTIFICIAL EYE Artificial Eye Market, BY REGION
- 8.1 INTRODUCTION
-
8.2 AMERICAS
-
8.2.1 NORTH AMERICA
- 8.2.1.2 U.S
- 8.2.1.2 CANADA
-
8.2.2 LATIN AMERICA
- 8.2.2.1 ARGENTINA
- 8.2.2.2 BRAZIL
- 8.2.2.3 MEXICO
- 8.2.2.4 REST OF LATAM
-
8.2.1 NORTH AMERICA
-
8.3 EUROPE
-
8.3.1 WESTERN EUROPE
- 8.3.1.1 GERMANY
- 8.3.1.2 FRANCE
- 8.3.1.3 ITALY
- 8.3.1.4 SPAIN
- 8.3.1.5 U.K
- 8.3.1.6 REST OF WESTERN EUROPE
-
8.3.2 EASTERN EUROPE
- 8.3.2.1 POLAND
- 8.3.2.2 RUSSIA
-
8.3.1 WESTERN EUROPE
-
8.4 ASIA-PACIFIC
- 8.4.1 JAPAN
- 8.4.2 CHINA
- 8.4.3 INDIA
- 8.4.4 REPUBLIC OF KOREA
- 8.4.5 AUSTRALIA
- 8.4.6 NEW ZEALAND
-
8.5 MIDDLE EAST & AFRICA
- 8.5.1 UAE
- 8.5.2 SAUDI AERBIA
- 8.5.3 QATAR
- 8.5.4 AFRICA
- 8.5.5 SOUTH AFRICA
- 8.5.6 REST OF AFRICA
-
9 COMPETITIVE LANDSCAPE
-
9.1 MAJOR STRATEGIES ADOPTED BY MARKET PLAYERS
- 9.1.1 STRATEGIC PARTNERSHIP
- 9.1.2 MERGER & ACQUISITION
-
9.1 MAJOR STRATEGIES ADOPTED BY MARKET PLAYERS
-
10 COMPANY PROFILE
-
10.1 ADVANCE ARTIFICIAL EYE
- 10.1.1 OVERVIEW
- 10.1.2 PRODUCT OVERVIEW
- 10.1.3 FINANCIALS
- 10.1.4 KEY DEVELOPMENTS
-
10.2 COS MEDIC PTV LTD
- 10.2.1 OVERVIEW
- 10.2.2 PRODUCT OVERVIEW
- 10.2.3 FINANCIALS
- 10.2.4 KEY DEVELOPMENTS
-
10.3 ERICKSON CUSTOM MADE ARTIFICIAL EYE
- 10.3.1 OVERVIEW
- 10.3.2 PRODUCT OVERVIEW
- 10.3.3 FINANCIALS
- 10.3.4 KEY DEVELOPMENT
-
10.4 INTERNATIONAL PROSTHETIC EYE CENTER
- 10.4.1 OVERVIEW
- 10.4.2 PRODUCT OVERVIEW
- 10.4.3 FINANCIALS
- 10.4.4 KEY DEVELOPMENTS
-
10.5 MARIE ALLEN OCULARIST LTD
- 10.5.1 OVERVIEW
- 10.5.2 PRODUCT OVERVIEW
- 10.5.3 FINANCIALS
- 10.5.4 KEY DEVELOPMENTS
-
10.6 THE NATIONAL ARTIFICIAL EYE SERVICES
- 10.6.1 OVERVIEW
- 10.6.2 PRODUCT OVERVIEW
- 10.6.3 FINANCIALS
- 10.6.4 KEY DEVELOPMENTS
- 10.7 OTHERS
-
10.1 ADVANCE ARTIFICIAL EYE
Customer Stories

“This is really good guys. Excellent work on a tight deadline. I will continue to use you going forward and recommend you to others. Nice job”

“Thanks. It’s been a pleasure working with you, please use me as reference with any other Intel employees.”

“Thanks for sending the report it gives us a good global view of the Betaïne market.”

“Thank you, this will be very helpful for OQS.”

“We found the report very insightful! we found your research firm very helpful. I'm sending this email to secure our future business.”

“I am very pleased with how market segments have been defined in a relevant way for my purposes (such as "Portable Freezers & refrigerators" and "last-mile"). In general the report is well structured. Thanks very much for your efforts.”

“I have been reading the first document or the study, ,the Global HVAC and FP market report 2021 till 2026. Must say, good info! I have not gone in depth at all parts, but got a good indication of the data inside!”

“We got the report in time, we really thank you for your support in this process. I also thank to all of your team as they did a great job.”

