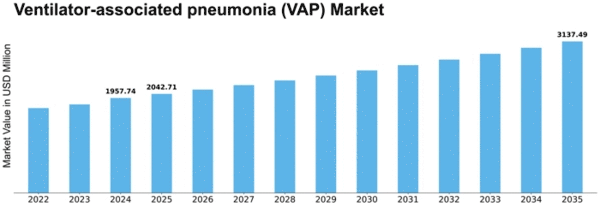
Ventilator Associated Pneumonia Vap Size
Ventilator-associated pneumonia (VAP) Market Growth Projections and Opportunities
The market for ventilator-associated pneumonia (VAP) treatment is significantly prompted by using hospitalization charges, especially in essential care units in which sufferers frequently require mechanical air flow. Higher fees for hospitalizations, mainly in intensive care settings, contribute to improved times of VAP, driving the demand for powerful remedies and preventive measures. The prevalence of VAP is immediately tied to the variety of patients receiving mechanical ventilation. As ventilator use becomes extra common in crucial care settings, the occurrence of VAP rises. This occurrence shapes the market for VAP-unique interventions and treatment options aimed toward lowering the risk of pneumonia in ventilated patients. The emergence of microbial resistance and the particular pathogen profile associated with VAP impact the market for antibiotic remedies. Antimicrobial resistance affects the development of recent antibiotics and alternative treatment procedures to combat VAP-inflicting pathogens efficiently. The development of prescription drugs, along with antibiotics and antifungal medicinal drugs, is a critical component of the VAP market. Ongoing research and improvements in drug formulations contribute to the availability of powerful remedies for VAP-inflicting pathogens, influencing market dynamics. Patient positioning and mobilization practices affect VAP prevention. Strategies to lessen the duration of mechanical ventilation, put into effect the right patient positioning, and inspire early mobilization make contributions to reducing the hazard of VAP, influencing market trends toward holistic care techniques. The training of healthcare workers in VAP prevention measures is essential. Education on exceptional practices, adherence to protocols, and early popularity of VAP signs contribute to improved patient results and have an impact on the market for schooling programs and educational assets. The excellent respiration care offerings in healthcare facilities are a big market aspect. Well-skilled respiratory therapists and specialized care groups make contributions to the prevention and early detection of VAP, impacting typical patient care and market alternatives. Global fitness-demanding situations, such as pandemics, can affect the VAP market. Surges in important care admissions and the want for mechanical air flow for the duration of health crises can also impact the superiority of VAP, affecting market tendencies and resource allocation. Adherence to regulatory recommendations and requirements set by using health authorities is crucial within the VAP market. Compliance with infection manipulation protocols, ventilator upkeep requirements, and reporting practices ensures patient safety and impacts market recognition of healthcare facilities and products.




















Leave a Comment