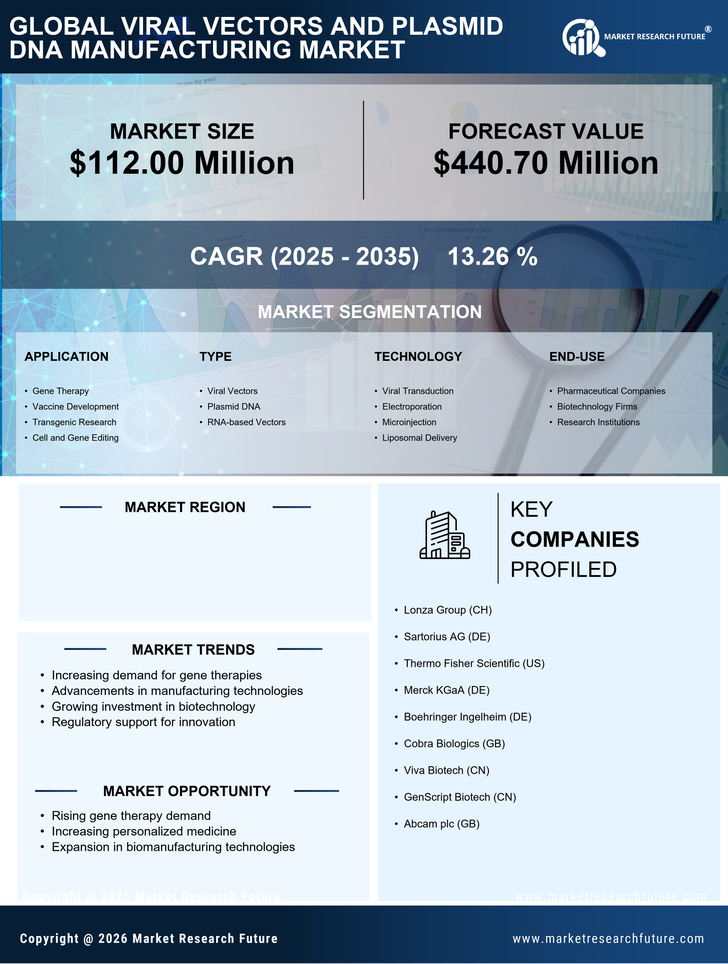
US Medical Device Market Segmentation
Medical Device By Application (USD Million, 2025-2035)
- Gene Therapy
- Vaccine Development
- Transgenic Research
- Cell and Gene Editing
Medical Device By Type (USD Million, 2025-2035)
- Viral Vectors
- Plasmid DNA
- RNA-based Vectors
Medical Device By Technology (USD Million, 2025-2035)
- Viral Transduction
- Electroporation
- Microinjection
- Liposomal Delivery
Medical Device By End-Use (USD Million, 2025-2035)
- Pharmaceutical Companies
- Biotechnology Firms
- Research Institutions