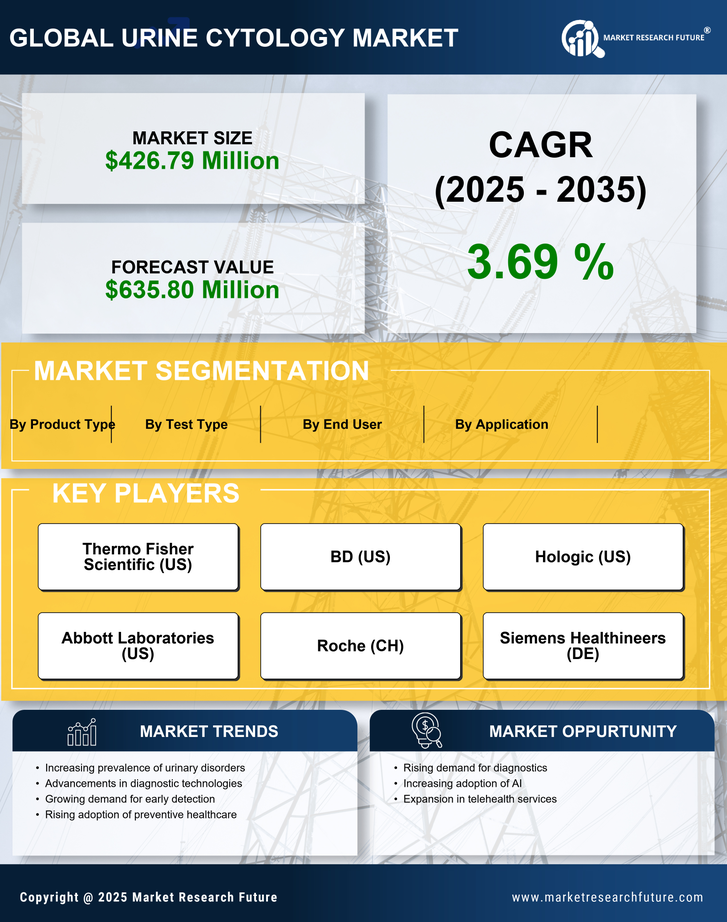
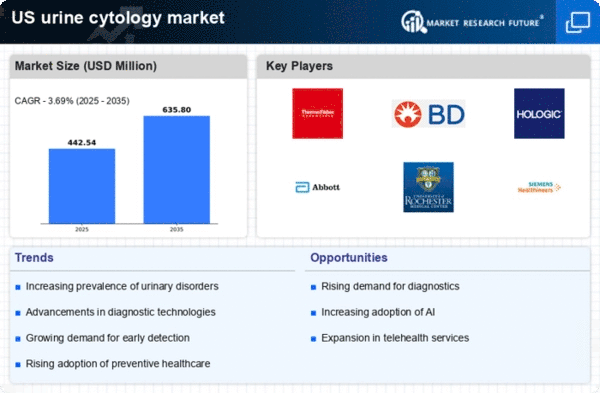
US Medical Device Market Segmentation
Medical Device By Test Type (USD Million, 2025-2035)
- Conventional Urine Cytology
- Automated Urine Cytology
- Fluorescence In Situ Hybridization
Medical Device By Application (USD Million, 2025-2035)
- Bladder Cancer Diagnosis
- Urinary Tract Infection Detection
- Kidney Disease Assessment
Medical Device By End User (USD Million, 2025-2035)
- Hospitals
- Clinical Laboratories
- Research Institutes
Medical Device By Product Type (USD Million, 2025-2035)
- Reagents
- Consumables
- Equipment