US Security, Access Control and Robotics Market Segmentation
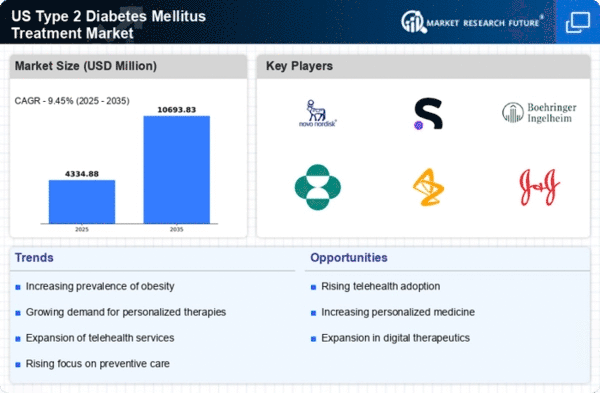
Security, Access Control and Robotics By Treatment (USD Million, 2025-2035)
- Drug Class
- Devices
Security, Access Control and Robotics By Drug Class (USD Million, 2025-2035)
- Insulin
- Sensitizers
- SGLT-2 Inhibitors
- Alpha-Glucosidase Inhibitors
- Secretagogues
- Peptide Analogs
- Others
Security, Access Control and Robotics By Devices (USD Million, 2025-2035)
- Blood Glucose Monitoring Devices
- Insulin Delivery Devices
Security, Access Control and Robotics By End User (USD Million, 2025-2035)
- Hospitals
- Clinics
- Diagnostic Centers
- Drug Stores
- Pharmacies
- Others
.png)